01
Native Conformation Recognition
Directly recognizes GPCRs in their native conformation on the live-cell membrane, accurately reflecting physiological receptor expression.
G protein-coupled receptors (GPCRs) represent the largest and most diverse family of receptors in the human body, playing essential roles in neurotransmission, immune regulation, endocrine signaling, and metabolic homeostasis. Currently, more than 30% of approved drugs directly or indirectly target GPCRs. With the rapid advances in research on metabolic disorders, cancer immunotherapy, and neurological diseases, an increasing number of orphan GPCRs have been identified as being closely associated with disease pathogenesis, further reinforcing GPCRs as one of the most promising and enduring classes of drug discovery targets.
However, the unique structural characteristics of GPCRs pose significant challenges for both basic research and drug development. As highly hydrophobic, multi-pass transmembrane proteins, GPCRs rely on an intact lipid membrane environment to maintain their native conformations. Once removed from the cell membrane, they are prone to conformational changes or even loss of function. Consequently, traditional detection methods based on denatured or fixed conditions—such as Western blotting, ELISA, or immunohistochemistry—often fail to accurately reflect GPCR expression on the surface of living cells or their physiological relevance. This limitation significantly hinders reliable receptor characterization, signaling mechanism studies, and the accurate screening of drug candidates.
To truly understand GPCR biology and advance drug development, it is essential to directly detect and quantify cell-surface receptors in their native conformation on living cells. By enabling analysis in an intact membrane environment, GPCR flow cytometry antibodies have become a critical tool for evaluating receptor expression, antibody binding, and target accessibility.
Addressing the conformational dependence of GPCRs and the need for live-cell analysis, flow cytometry antibodies deliver the following core values in research and drug discovery:
Directly recognizes GPCRs in their native conformation on the live-cell membrane, accurately reflecting physiological receptor expression.
Provides quantitative measurements at single-cell resolution, enabling precise comparison of GPCR expression under different conditions.
Selects only antibodies that stably bind GPCRs on the surface of living cells, ensuring robust, reproducible flow cytometry signals.
Assesses target accessibility and antibody developability, reducing early-stage risk in GPCR-targeted drug discovery.
Leveraging the ability to directly detect GPCRs on living cells by flow cytometry, GPCR flow cytometry antibodies demonstrate significant advantages in both research and development. By preserving native conformation recognition, enabling quantitative single-cell analysis, integrating seamlessly with multiple functional assays, and supporting diverse cell types, these antibodies provide a more physiologically relevant approach for GPCR expression validation, antibody screening, and functional studies.
The figure below summarizes the key advantages of GPCR flow cytometry antibodies across major application scenarios.
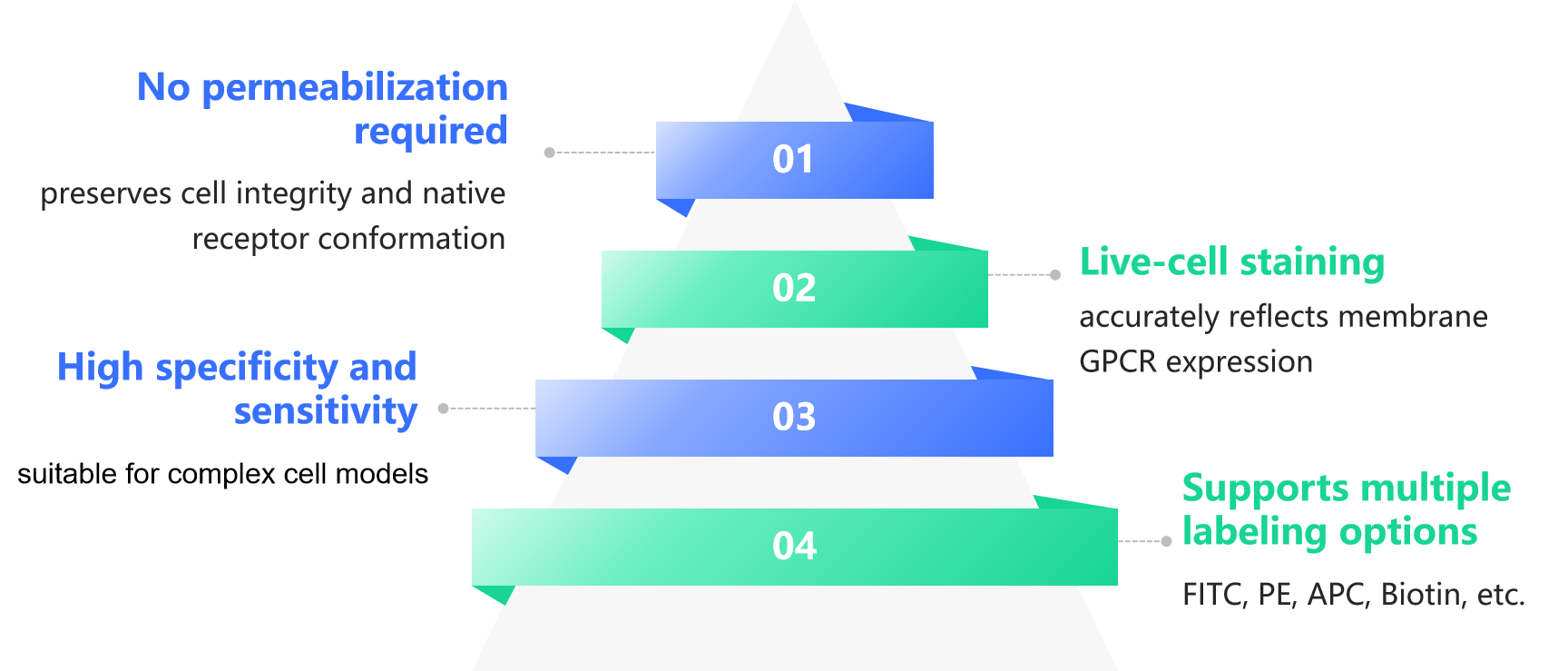
Antibodies recognize receptors in their native state on the surface of living cells, without relying on denaturation or fixation, thereby more accurately reflecting physiological conditions.
Can be combined with Ca²⁺ flux assays, signaling pathway analyses, and internalization studies to establish a comprehensive GPCR research workflow.
Flow cytometry enables precise quantification at single-cell resolution, facilitating direct comparison of GPCR expression levels across different treatment conditions.
Suitable for rapid assessment of receptor distribution in primary cells, transfected cell lines, and heterogeneous immune cell populations.
GPCRs are complex seven-transmembrane proteins whose native conformations are difficult to preserve with traditional antibodies, which often require permeabilization and can disrupt membranes, cause signal loss, or introduce false positives. DIMA’s GPCR flow cytometry antibodies overcome these limitations by using native-conformation antigens generated through our DiMProTM Nanodisc platform. These antibodies specifically recognize GPCR epitopes on the surface of live cells, enabling direct detection without permeabilization. This approach allows researchers to measure GPCR expression and conformational changes under conditions that closely reflect physiological states, providing more reliable, accurate, and biologically meaningful data for GPCR research and drug development.
Key advantages:
DIMA offers an extensive library of GPCR flow cytometry antibodies, covering multiple research areas including immunology, neuroscience, metabolism, and oncology:
| Target | Cat.No. | Product Name | Applications |
| CB1 | DME100144 | Anti-CB1 antibody(DM144); Rabbit mAb | ELISA; Flow Cyt |
| GIPR | DMC101001 | Anti-GIPR antibody(3G5); IgG1 Chimeric mAb | Flow Cyt |
| GLP1R | DME101291 | Anti-GLP1R antibody(1A4), Rabbit mAb | Flow Cyt |
| GPR75 | DMC100368 | Anti-GPR75 antibody(DMC368); IgG1 Chimeric mAb | Flow Cyt |
| GPR75 | DMC100492 | Anti-GPR75 antibody(DMC492); IgG1 Chimeric mAb | Flow Cyt |
| GPR75 | DMC100368B | Biotinylated Anti-GPR75 antibody(DMC368); IgG1 Chimeric mAb | Flow Cyt |
| GPR75 | DMC100492B | Biotinylated Anti-GPR75 antibody(DMC492); IgG1 Chimeric mAb | Flow Cyt |
| GPR75 | DME101136 | Anti-GPR75 antibody(12B6), Rabbit mAb | Flow Cyt |
| GPR75 | DME101136B | Biotinylated Anti-GPR75 antibody(12B6), Rabbit mAb | Flow Cyt |
| GPR75 | DMC100368P | PE-conjugated Anti-GPR75 antibody(DMC368); IgG1 Chimeric mAb | Flow Cyt |
| LGR4 | DME101188 | Anti-LGR4 antibody(1D9), Rabbit mAb | Flow Cyt |
| MC4R | DME101294 | Anti-MC4R antibody(2H10), Rabbit mAb | Flow Cyt |
| TSHR | DMC101228 | Anti-TSHR antibody(1C8), IgG1 Chimeric mAb | Flow Cyt |
| Target | Cat.No. | Product Name | Applications |
| CB1 | DME100144 | Anti-CB1 antibody(DM144); Rabbit mAb | ELISA; Flow Cyt |
| GPR20 | DME101292 | Anti-GPR20 antibody(1E10), Rabbit mAb | Flow Cyt |
| GRPR | DME101290 | Anti-GRPR antibody(3B12), Rabbit mAb | Flow Cyt |
| MC4R | DME101294 | Anti-MC4R antibody(2H10), Rabbit mAb | Flow Cyt |
| SSTR2 | DMC100428 | Anti-SSTR2 antibody(DMC428); IgG1 Chimeric mAb | Flow Cyt |
…More GPCR targets are continuously being added.
Powered by our DiMProTM Nanodisc , DIMA is steadily expanding its portfolio of GPCR flow cytometry antibodies that recognize native conformations. These tools enable researchers to perform high-specificity live-cell detection and quantitative analysis with ease, providing comprehensive support for GPCR drug discovery.
Our GPCR antibodies are carefully developed and validated to target native extracellular epitopes, ensuring high specificity in live-cell flow assays.
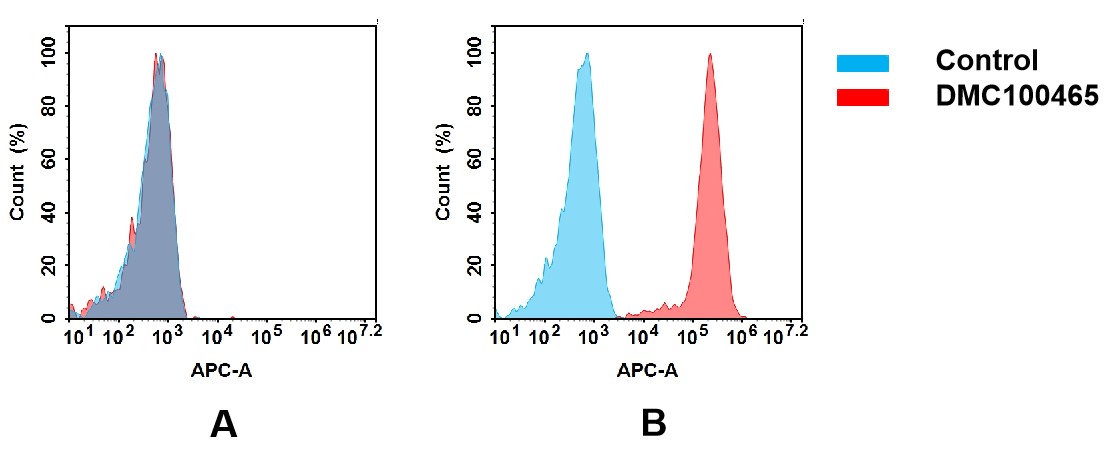
(A) DMC100465 does not bind to CHO-S cells that do not express CCR1.
(B) A clear peak shift of DMC100465 was seen compared to the control when incubated with CCR1-expressing 8226 cells, indicating strong binding of DMC100465 to CCR1. Antibodies were incubated at 5 μg/ml.
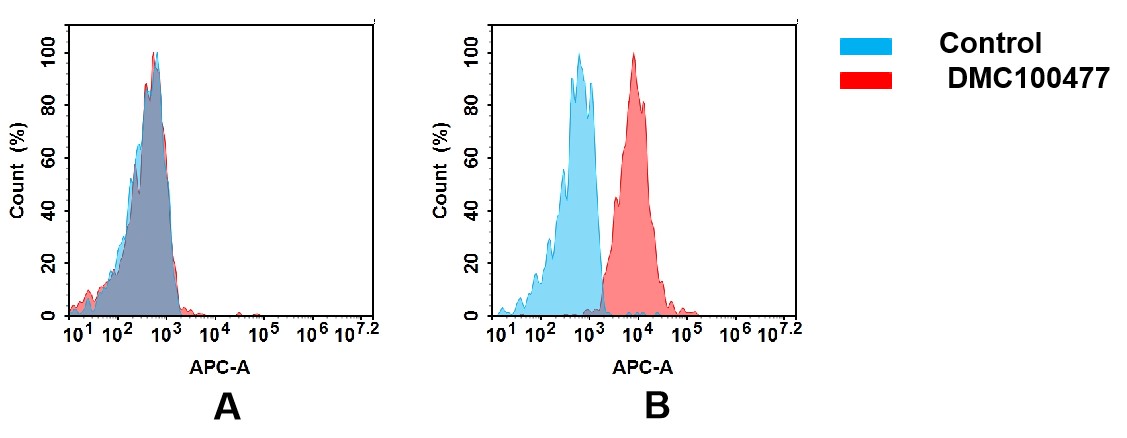
(A) DMC100477 does not bind to CHO-S cells that do not express CCR6.
(B) A clear peak shift of DMC100477 was seen compared to the control when incubated with CCR6-expressing Huh7 cells, indicating strong binding of DMC100477 to CCR6. Antibodies were incubated at 5 μg/mL.
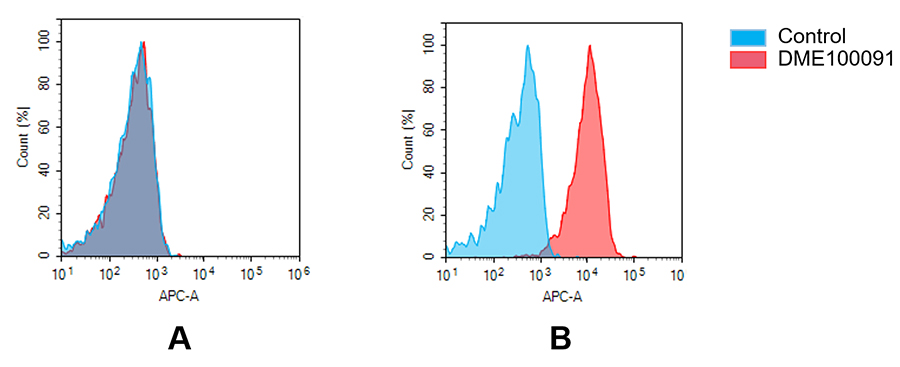
(A) DME100091 does not bind to Jurkat cells that do not express GPRC5D.
(B) A clear peak shift of DME100091 was seen compared to the control when incubated with GPRC5D-expressing MM.1S cells, indicating strong binding of DME100091 to GPRC5D. Antibodies were incubated at 5 µg/mL.