At the 2025 American Society of Clinical Oncology (ASCO) Annual Meeting, CCR8 re-emerged as a major focus in immuno-oncology, positioning it as a next-generation strategy for precision Treg depletion.For example, LM-108 (Cafelkibart), developed by LaNova Medicines in collaboration with Sino Biopharmaceutical, showed encouraging efficacy in advanced gastric cancer and has advanced into Phase III. ICP-B05 from Innocare Pharma achieved a 72.9% 6-month PFS rate in CTCL. Meanwhile, high-value deals between Bristol Myers Squibb and Agenus, as well as Gilead Sciences’ acquisition of Jounce’s CCR8 program, further validate the target’s commercial potential.
With selective expression on tumor-infiltrating Tregs and minimal peripheral presence, CCR8 enables precise intratumoral Treg depletion with reduced systemic toxicity, making it a promising strategy beyond PD-1/L1 resistance.
1. CCR8 Structure and Tissue Distribution
Chemokine receptor 8 (CCR8) is a Class A G protein-coupled receptor (GPCR) composed of 355 amino acids with a canonical seven-transmembrane (7TM) structure. Its extracellular N-terminus plays a role of ligand recognition; and its intracellular C-terminus is responsible for G-protein coupling and signal transduction.
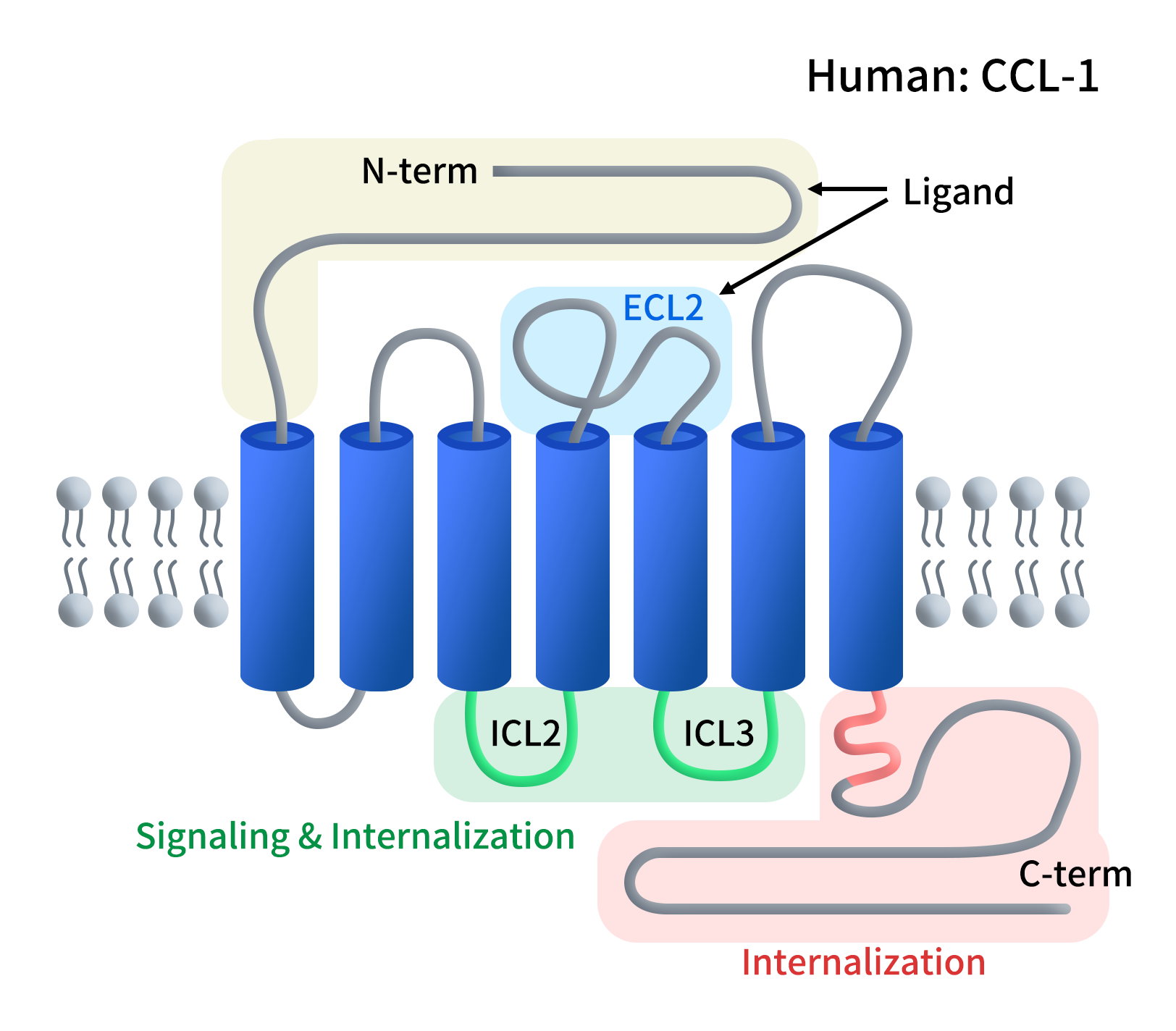
Figure 1. The structure of CCR8
CCR8 expression is highly selective, which is expressed on Th2 cells, monocytes, and NK cells, not expressed on Th1 cells, and minimal expression on peripheral Tregs. Note that, CCR8 is highly enriched on tumor-infiltrating Tregs (TITRs), which has been reported in breast cancer, lung cancer, colorectal cancer, and gastric cancer, reinforcing its tumor-specific relevance.
2.CCR8 Function and Disease Association
The physiological Role of CCR8 is recruitment of Th2 cells, and maintenance of skin and mucosal immune homeostasis. But, In cancer, CCR8 interacts primarily with its ligand CCL1, driving recruitment of FOXP3+ CCR8+ Tregs into tumors, upregulation of Foxp3, CD39, IL-10, Granzyme B via STAT3 signaling, enhanced immunosuppressive function, and formation of a barrier blocking CD8+ cytotoxic T-cell infiltration. This mechanism makes the CCR8–CCL1 axis a central driver of tumor immune suppression. Additionally, CCR8 is also implicated in atopic dermatitis and asthma (via Th2 recruitment), and potential fibrosis-related macrophage recruitment.
3. CCR8 Ligands and Tumor Signaling Pathways
Known human CCR8 ligands include CCL1 (dominant functional ligand), CCL8, CCL16 and CCL18. Among them, CCL1 is the principal and functionally dominant ligand. Human CCL1 binds to both the extracellular loop 2 (ECL2) and the N-terminal domain of CCR8. Although CCR8 can also interact with CCL8, CCL16, and certain viral chemokines, the CCL1/CCR8 axis is the central driver of Treg migration and immunosuppressive function within the tumor microenvironment.
3.1 CCL1/CCR8 Axis in Tumors
CCR8 is highly expressed on tumor-infiltrating regulatory T cells (Tregs), while CCL1 is primarily produced by cancer-associated fibroblasts (CAFs), tumor-associated macrophages (TAMs), tumor-infiltrating Tregs, and certain cancer stem cells.
Within the tumor microenvironment (TME), CCL1 activates CCR8 expressed on cancer cells, promoting tumor cell proliferation, resistance to apoptosis, and enhanced migratory capacity. Beyond its direct effects on tumor cells, CCL1 also stimulates CCR8 on endothelial cells, thereby contributing to angiogenesis.
A key immunological function of CCL1 is the recruitment of Tregs into the tumor niche. In addition, CCL1 participates in the conversion of CD4⁺ T cells into Tregs, a process that depends on transforming growth factor-β (TGF-β). Through these mechanisms, CCL1 reinforces the immunosuppressive activity of Tregs within the tumor ecosystem.
Given its multifaceted role in tumor progression and immune evasion, targeting the CCL1–CCR8 axis has emerged as a novel immune checkpoint-based therapeutic strategy in oncology.
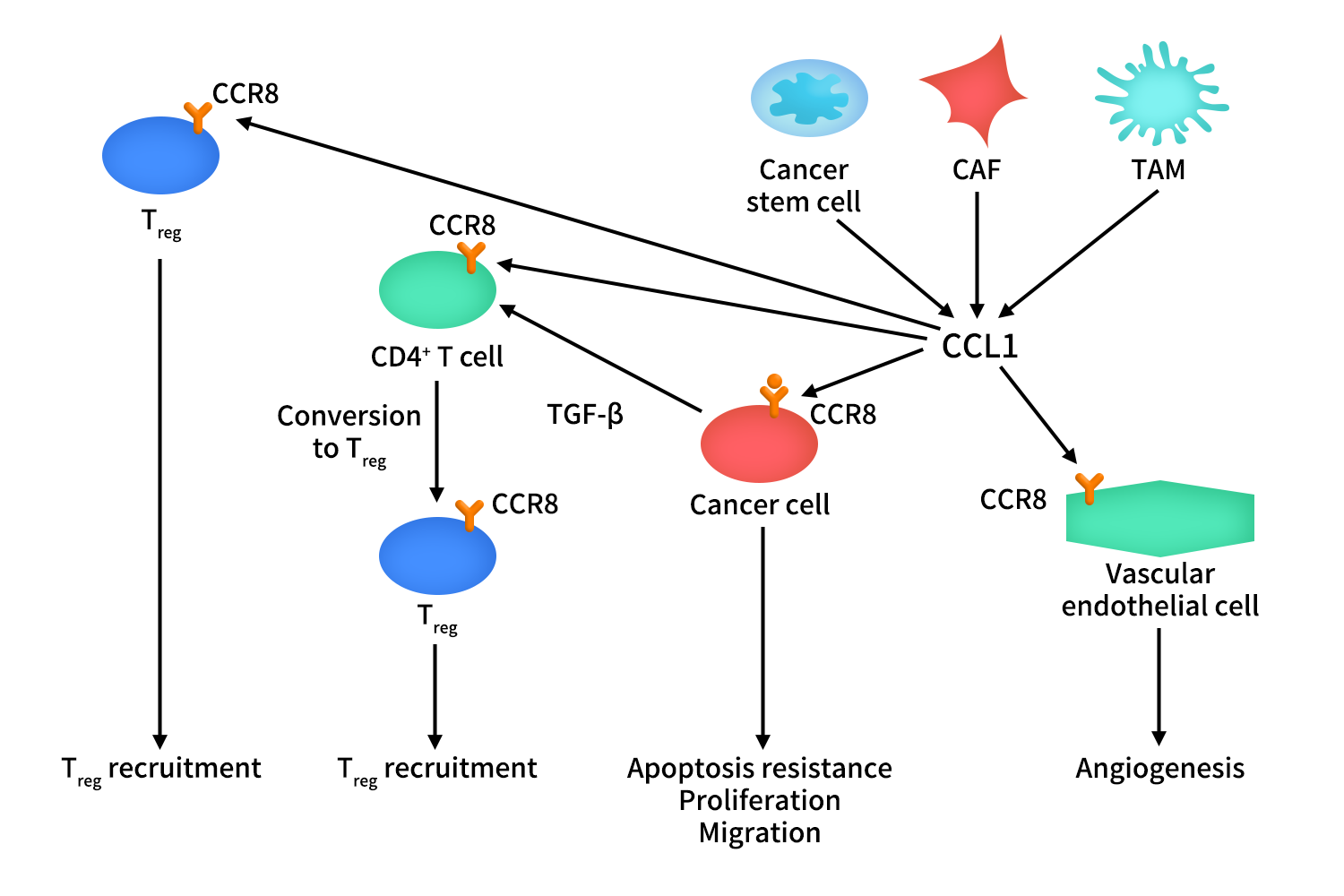
Figure 2. The mechanism of CCR8-CCL1 axis in cancer [1]
3.2 CCL18/CCR8 Axis in Cancer
In tumors, CCL18 is mainly produced and secreted by TAMs and can act in an autocrine manner on these cells. CCL18 promotes cancer cell migration and invasion through its receptors PITPNM3 and CCR8. It also plays an important role in angiogenesis by acting on endothelial cells via PITPNM3.
Furthermore, CCL18 facilitates the recruitment of naïve CD4⁺ T cells into the tumor niche and promotes their differentiation into Treg cells, thereby contributing to the establishment and maintenance of an immunosuppressive tumor microenvironment.
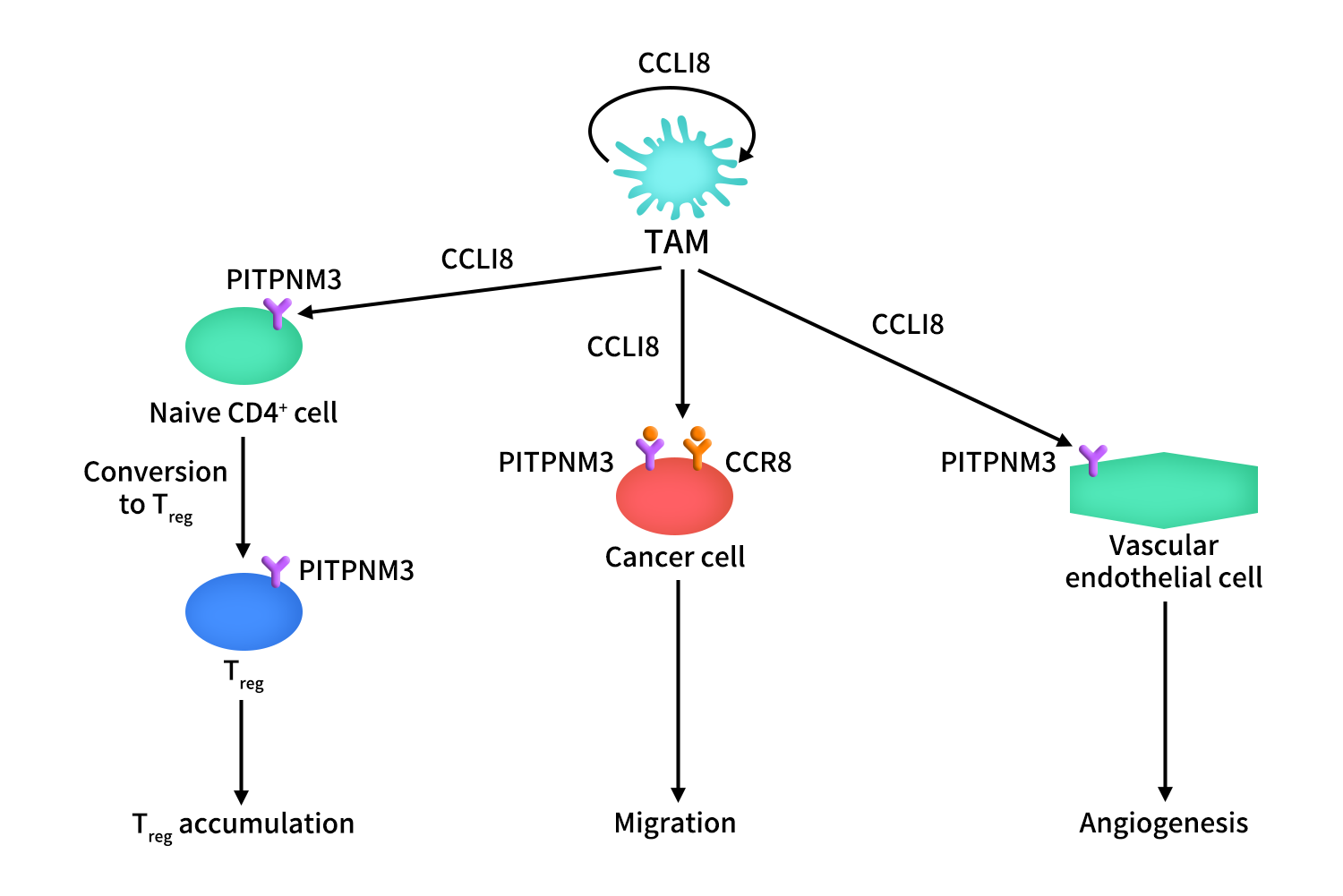
Figure 3. The mechanism of CCR8-CCL18 axis in cancer [1]
4. Clinical Progress of CCR8-Targeted Therapies
The highly selective overexpression of CCR8 on Tregs underscores its value as a robust biomarker of intratumoral Tregs and a highly promising immuno-oncology target. As a result, CCR8 has become a major focus of global drug development efforts. According to incomplete statistics, there are currently 73 CCR8-targeted programs worldwide. Among these, 23 are in the preclinical stage, 16 are in the discovery stage, 11 have entered Phase I clinical trials, and Only 4 programs have advanced to Phase II or Phase III.
The dominant therapeutic modalities are monoclonal antibodies and small-molecule antagonists, although a limited number of bispecific antibodies, antibody–drug conjugates (ADCs), and CAR-T programs are also in development. The following section highlights key monoclonal antibody and small-molecule programs.
4.1 CCR8 Monoclonal Antibodies
Currently, 14 CCR8 monoclonal antibodies have entered clinical development, with only one reaching Phase III; most remain in early-stage (Phase I) trials.
- LM-108 (Cafelkibart), developed by LaNova Medicines, is a humanized anti-CCR8 monoclonal antibody designed to selectively deplete CCR8-expressing Tregs in the tumor microenvironment through antibody-dependent cellular cytotoxicity (ADCC) and antibody-dependent cellular phagocytosis (ADCP). By reducing intratumoral immunosuppression, LM-108 aims to restore and enhance anti-tumor immune responses.LM-108 has initiated a Phase III registration trial (NCT07362186) as second-line therapy for CCR8-positive locally advanced or metastatic gastric/gastroesophageal junction (GEJ) adenocarcinoma. In this randomized controlled study conducted in China, LM-108 combined with toripalimab is being compared with paclitaxel. This marks the most advanced clinical stage reached by any CCR8-targeted therapy to date.
- BMS-986340 (Imzokitug), developed by Bristol Myers Squibb, is an afucosylated humanized IgG1 anti-CCR8 antibody. The afucosylation design enhances Fc receptor binding to effector cells, thereby significantly increasing ADCC activity and improving the selective depletion of CCR8-positive tumor-infiltrating Tregs. The goal is to remodel the tumor immune microenvironment and activate anti-tumor immunity.BMS-986340 is currently being evaluated in global Phase I/II trials, both as monotherapy and in combination with PD-1 inhibitors. These studies aim to assess safety, tolerability, and preliminary efficacy. The program is presently in dose-escalation and patient recruitment stages.
- S-531011, developed by Shionogi is a humanized IgG1 monoclonal antibody that specifically targets CCR8. It possesses strong ADCC activity and the ability to block CCR8/CCL1 signaling, thereby attenuating the immunosuppressive function of CCR8-positive Tregs within the tumor microenvironment and restoring anti-tumor immune responses.In preclinical models, S-531011 demonstrated the ability to reduce tumor-infiltrating Tregs and significantly inhibit tumor growth. Combination therapy with PD-1 inhibitors in mouse models produced enhanced anti-tumor effects, with no severe adverse events observed. The agent is currently advancing through Phase 1b/2 clinical trials in Japan and the United States to evaluate safety, recommended dose, and preliminary efficacy.
- GS-1811 (Denikitug), originally developed by Jounce Therapeutics, is an afucosylated humanized monoclonal antibody targeting CCR8. It is designed to selectively eliminate immunosuppressive tumor-infiltrating Tregs that highly express CCR8. Through enhanced ADCC activity, GS-1811 blocks CCR8/CCL1 signaling and induces apoptosis of CCR8-positive Tregs, thereby aiming to relieve immune suppression and stimulate anti-tumor immunity.The drug is currently being evaluated in a first-in-human Phase I clinical trial (NCT05007782), assessing its safety, tolerability, maximum tolerated dose (MTD), and recommended Phase II dose both as monotherapy and in combination with the anti-PD-1 antibody zimberelimab in patients with advanced solid tumors. The trial remains ongoing and actively recruiting. In 2022, Gilead Sciences acquired the remaining development rights to this CCR8 antibody program, further underscoring the strategic importance of the target in immuno-oncology.
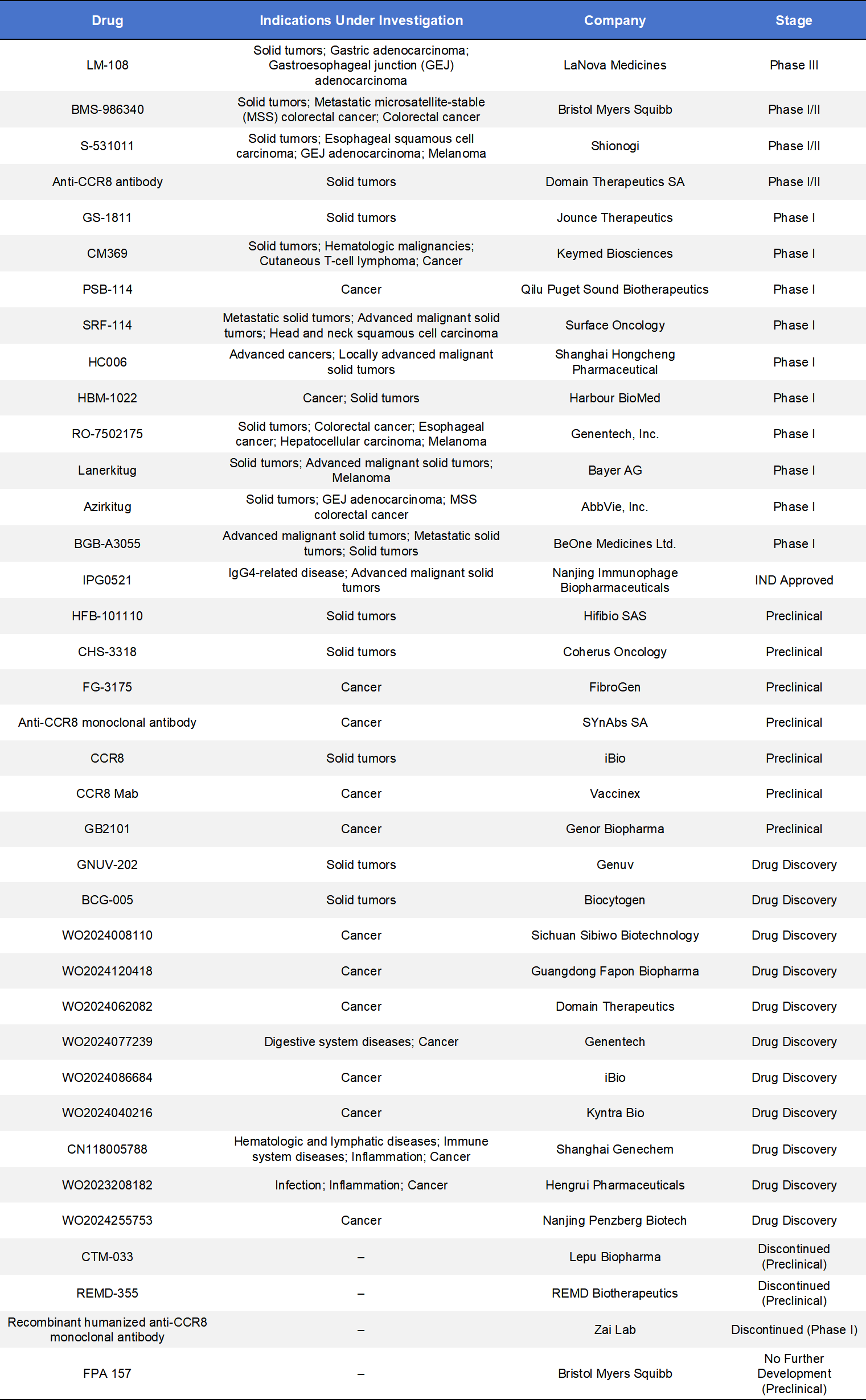
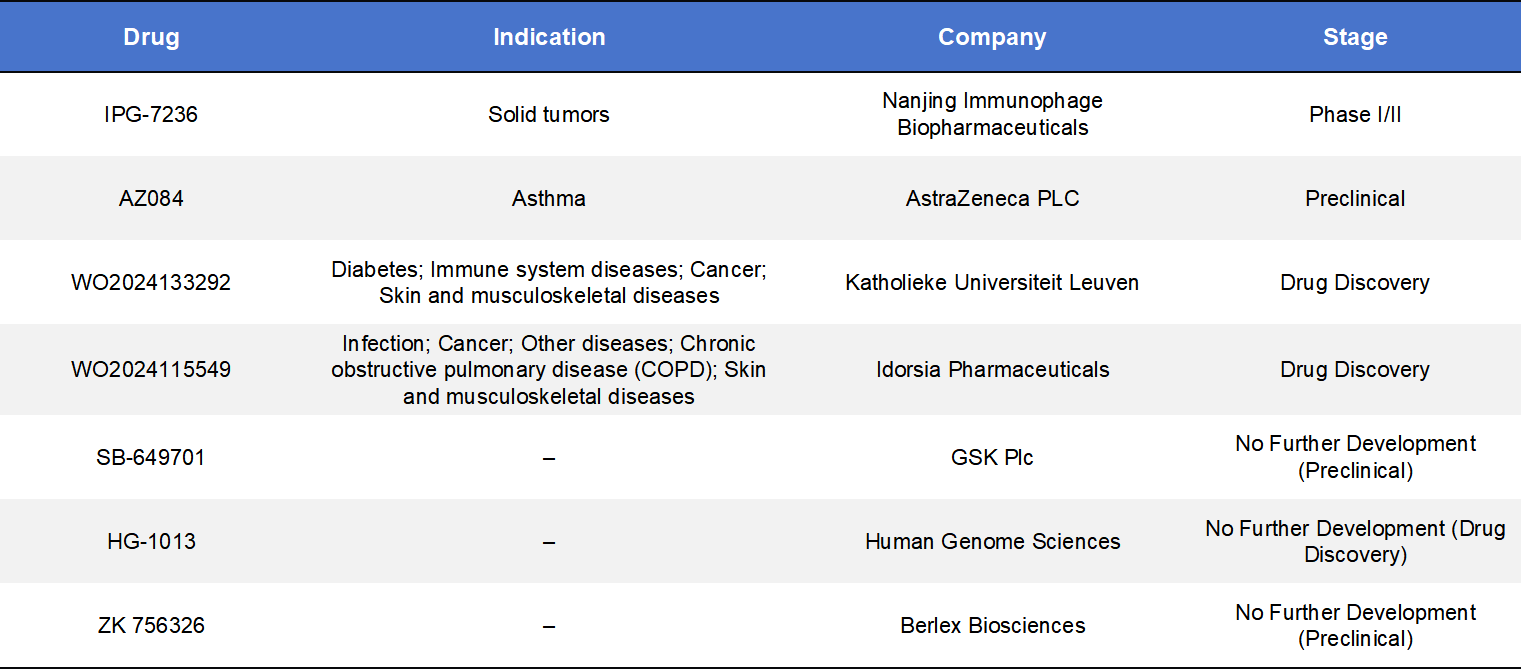
4.2 CCR8 Small-Molecule Antagonists
There are currently seven CCR8 small-molecule programs that have progressed into clinical development stages, with only one having entered clinical trials: IPG-7236, developed by Nanjing Immunophage Biopharmaceuticals.
IPG-7236 is an orally available small-molecule antagonist designed to disrupt the CCL1–CCR8 axis, thereby interfering with Treg recruitment into the tumor microenvironment. As a small molecule, it offers advantages such as improved tissue penetration and greater dosing convenience compared with antibody-based therapies. IPG-7236 has advanced into Phase I/II clinical studies. A multicenter, open-label dose-escalation and expansion trial is currently underway to evaluate its safety, tolerability, pharmacokinetics, and preliminary anti-tumor activity in patients with advanced solid tumors. The study includes multiple solid tumor subtypes, such as renal cell carcinoma, triple-negative breast cancer (TNBC), head and neck cancers, and melanoma.
In addition, Nanjing Immunophage has established a strategic collaboration with Shanghai GoBroad Cancer Hospital to accelerate the transition of IPG-7236 from Phase I to Phase II development and to further advance its clinical validation.
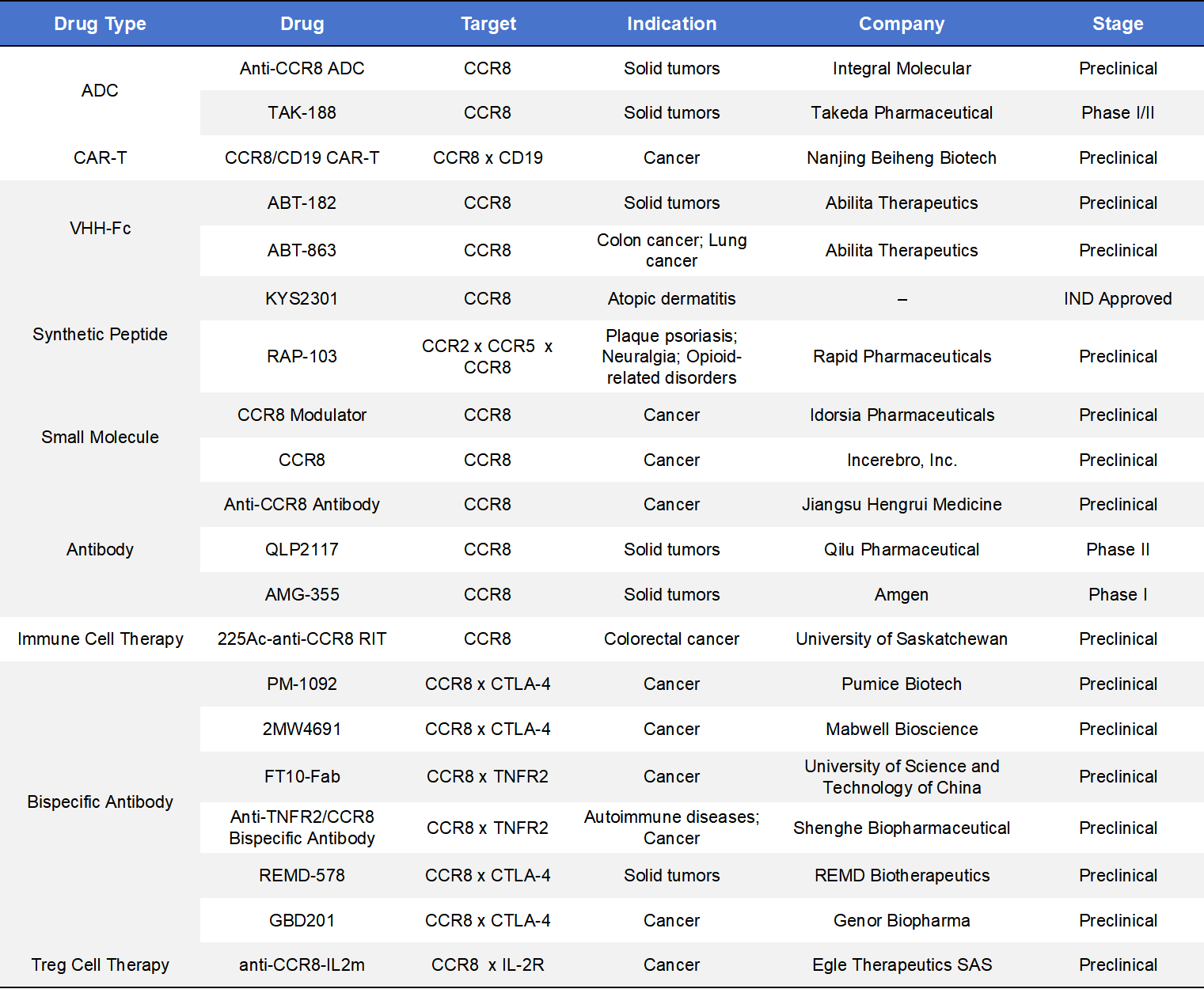
4.3 Other Modalities Targeting CCR8
Beyond monoclonal antibodies and small-molecule inhibitors, additional therapeutic approaches targeting CCR8 include antibody–drug conjugates (ADCs), CAR-T therapies, chemically synthesized drugs, and bispecific antibodies. Among these, bispecific antibodies represent the largest category in development, although most remain at the preclinical stage. To date, three CCR8-targeted agents have entered clinical development: TAK-188 (Takeda), QLP2117 (Qilu Pharmaceutical), and AMG-355 (Amgen).
- TAK-188 is an ADC developed by Takeda Pharmaceutical Company targeting CCR8. Upon binding to CCR8-expressing cells, TAK-188 delivers a cytotoxic payload directly to CCR8⁺ cells, selectively eliminating immunosuppressive Tregs within the tumor microenvironment (TME) and thereby enhancing antitumor immune responses. Depletion of CCR8⁺ Tregs is expected to restore and amplify CD8⁺ T-cell–mediated tumor killing. TAK-188 is currently being evaluated in a Phase I/II dose-escalation and expansion study in patients with locally advanced or metastatic solid tumors who have progressed after standard therapies. Multiple cohorts are included, such as head and neck squamous cell carcinoma and non-small cell lung cancer, assessing safety, tolerability, and preliminary efficacy.
- QLP2117, developed by Qilu Pharmaceutical, is an IgG1 monoclonal antibody targeting CCR8. It mediates antibody-dependent cellular cytotoxicity (ADCC) to deplete CCR8⁺ regulatory T cells in the TME, thereby relieving immunosuppression and promoting antitumor immune responses. QLP2117 is currently undergoing Phase I/II clinical studies, including a Phase I monotherapy safety trial and a Phase Ib/II dose-expansion study in combination with QL2107. These trials aim to evaluate safety, tolerability, pharmacokinetics, and preliminary efficacy in patients with advanced solid tumors.
- AMG-355, developed by Amgen, is a CCR8-targeted antibody designed to bind CCR8 in the tumor microenvironment, block the CCR8–CCL1 signaling axis, and selectively eliminate CCR8⁺ Tregs, thereby enhancing antitumor immunity. CCR8 is highly expressed on tumor-infiltrating Tregs and is strongly associated with immune suppression. AMG-355 is currently in a first-in-human Phase I study evaluating its safety, tolerability, and recommended Phase II dose, both as monotherapy and in combination with pembrolizumab (a PD-1 inhibitor), in patients with advanced solid tumors such as colorectal cancer, lung cancer, and gastric cancer.
5. CCR8-Related Products and Services from DIMA BIOTECH
DIMA BIOTECH is a biotechnology company specializing in preclinical research products and services for druggable targets. The company offers a comprehensive portfolio of CCR8-related products and solutions to support biologics development. Available products include active recombinant proteins, reference antibodies, and flow cytometry–validated monoclonal antibodies. Service capabilities encompass multi-species protein and antibody customization, antibody humanization, and affinity maturation.
To accelerate the development of CCR8-targeted biologics, DIMA BIOTECH has established a CCR8-specific single B-cell seed library, enabling lead antibody generation within as little as 20 days. Multiple CCR8 lead candidates have already been identified, with certain molecules available for functional evaluation as early as the next day upon request. For selected candidates, internalization activity and cytotoxicity validation for ADC development are currently underway. Detailed data are available upon inquiry.
|
Product Type |
Cat. No. |
Product Name |
|
ECD Protein |
PME101103 |
|
|
|
PME101091 |
|
|
|
PME-C100006 |
|
|
|
PME-M100034 |
|
|
The Full-length Protein |
FLP100001 |
|
|
|
FLP100037 |
|
|
FC-validated mAb |
DME100180 |
|
|
|
DME100195 |
|
|
|
DME100196 |
|
|
|
DME100197 |
|
|
Biosimilar mAb |
BME100063 |
|
|
|
BME100115 |
|
|
Biotin-labeled mAb |
DME100180B |
|
|
|
DME100195B |
|
|
|
DME100196B |
|
|
|
DME100197B |
|
|
|
BME100063B |
|
|
|
BME100115B |
Reference:
[1] Korbecki J , Grochans S , Gutowska I , et al. Cc chemokines in a tumor: A review of pro-cancer and anti-cancer properties of receptors ccr5, ccr6, ccr7, ccr8, ccr9, and ccr10 ligands[J]. International Journal of Molecular Sciences, 2020, 21(20):1-34.