In July 2025, Roche released updated clinical data on trontinemab, reigniting interest in blood–brain barrier (BBB) delivery strategies. Trontinemab is an engineered bispecific antibody: one arm targets pathogenic proteins (such as amyloid-β), while the other binds to Transferrin Receptor 1 (TfR1). By leveraging TfR1-mediated transport, trontinemab can actively cross the BBB from circulation into the brain, significantly improving central nervous system exposure and therapeutic efficacy.
Updated clinical results show that at a dose of 3.6 mg/kg, approximately 91% of patients achieved amyloid PET negativity after 28 weeks, with an ARIA-E incidence below 5%. The molecule has now advanced into Phase III clinical trials (TRONTIER 1/2), marking the first large-scale clinical validation of TfR1-mediated brain delivery. This milestone positions TfR1 not only as a key player in iron metabolism but also as one of the most promising translational gateways for CNS drug delivery.
1. TfR1 and the Transferrin Receptor Family
The transferrin receptor (TfR) family primarily consists of two subtypes: Transferrin Receptor 1 (TfR1) and Transferrin Receptor 2 (TfR2). Among them, TfR1 is the most widely expressed and extensively studied, whereas TfR2 is mainly involved in hepatic iron homeostasis and systemic iron regulation, with more tissue-specific functions.
TfR1, also known as CD71 and encoded by the TFRC gene, is located on chromosome 3q29. The TFRC gene spans approximately 30kb and contains multiple exons and introns. It encodes a polypeptide of 760 amino acids with a molecular weight of approximately 85–95 kDa.
Structurally, TfR1 is a classical type II transmembrane glycoprotein composed of an N-terminal cytoplasmic domain, a single transmembrane region, and a large C-terminal extracellular domain. The N-terminal intracellular region contains critical endocytosis signaling motifs that mediate clathrin-dependent internalization, forming the functional basis of iron uptake. The transmembrane domain anchors the receptor within the cell membrane and maintains its orientation. The extracellular domain, which constitutes the majority of the protein, forms a stable homodimer and contains the transferrin-binding sites responsible for ligand recognition and binding.
Structural studies show that two TfR1 monomers are linked via disulfide bonds formed by Cys89 and Cys98, ensuring dimer stability and efficient ligand binding. Additionally, TfR1 can undergo proteolytic cleavage at specific sites (such as near Arg100), releasing its extracellular domain into circulation as soluble TfR1 (sTfR). This dual existence, membrane-bound and soluble, makes TfR1 not only essential for iron metabolism but also a valuable biomarker with diagnostic and regulatory significance[1][2].
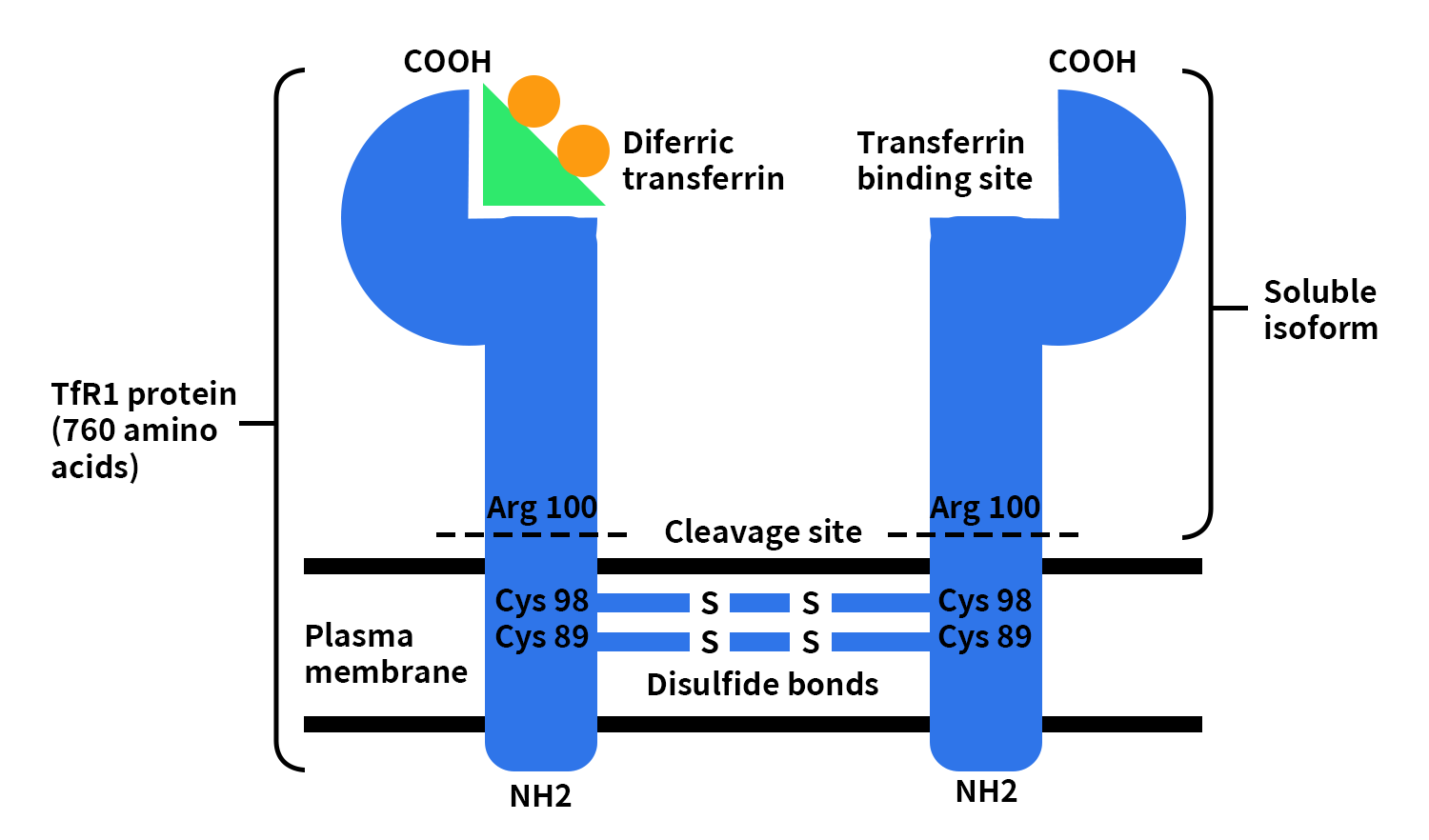
Figure 1. The picture of TfR1 structure [3]
2. Expression and Physiological Functions of TfR1
TfR1 is widely expressed across multiple cell types, with particularly high levels in cells and tissues that have elevated iron demand—such as erythroid precursor cells, tumor cells, and brain microvascular endothelial cells. Its primary biological role is to maintain intracellular iron homeostasis [2].
TfR1 binds to iron-loaded transferrin (holo-transferrin) and mediates receptor-dependent endocytosis, enabling efficient iron uptake into cells. The internalized iron is then utilized in essential biological processes, including DNA synthesis and mitochondrial metabolism [2] [4].
A key functional feature of TfR1 is its rapid recycling capability. After each endocytic cycle, the receptor quickly returns to the cell surface to participate in subsequent rounds of transport. This “high-throughput recycling system” significantly enhances transport efficiency and is one of the main reasons TfR1 has become an attractive target for drug delivery strategies—especially for large biomolecules.
3. Regulatory Mechanisms of TfR1 at the Blood–Brain Barrier (BBB)
Within the BBB, TfR1 is highly expressed on the luminal (blood-facing) side of brain microvascular endothelial cells. Beyond its central role in iron metabolism, TfR1 also functions as a critical gateway for molecular transport from the bloodstream into the brain.
Under physiological conditions, TfR1 first binds transferrin carrying ferric iron (Fe³⁺) and undergoes receptor-mediated endocytosis into endothelial cells. Inside endosomes, the acidic environment facilitates the reduction of Fe³⁺ to Fe²⁺, which is then released and transported into the cytoplasm via Divalent Metal Transporter 1 (DMT1). Subsequently, iron is exported toward the brain side through ferroportin, oxidized back to Fe³⁺, and reloaded onto transferrin in the brain interstitial fluid, completing the trans-BBB iron delivery process. Meanwhile, apo-transferrin (iron-free transferrin) remains bound to TfR1 and is recycled back to the cell surface, where it dissociates under neutral pH conditions, allowing the receptor to be reused efficiently.
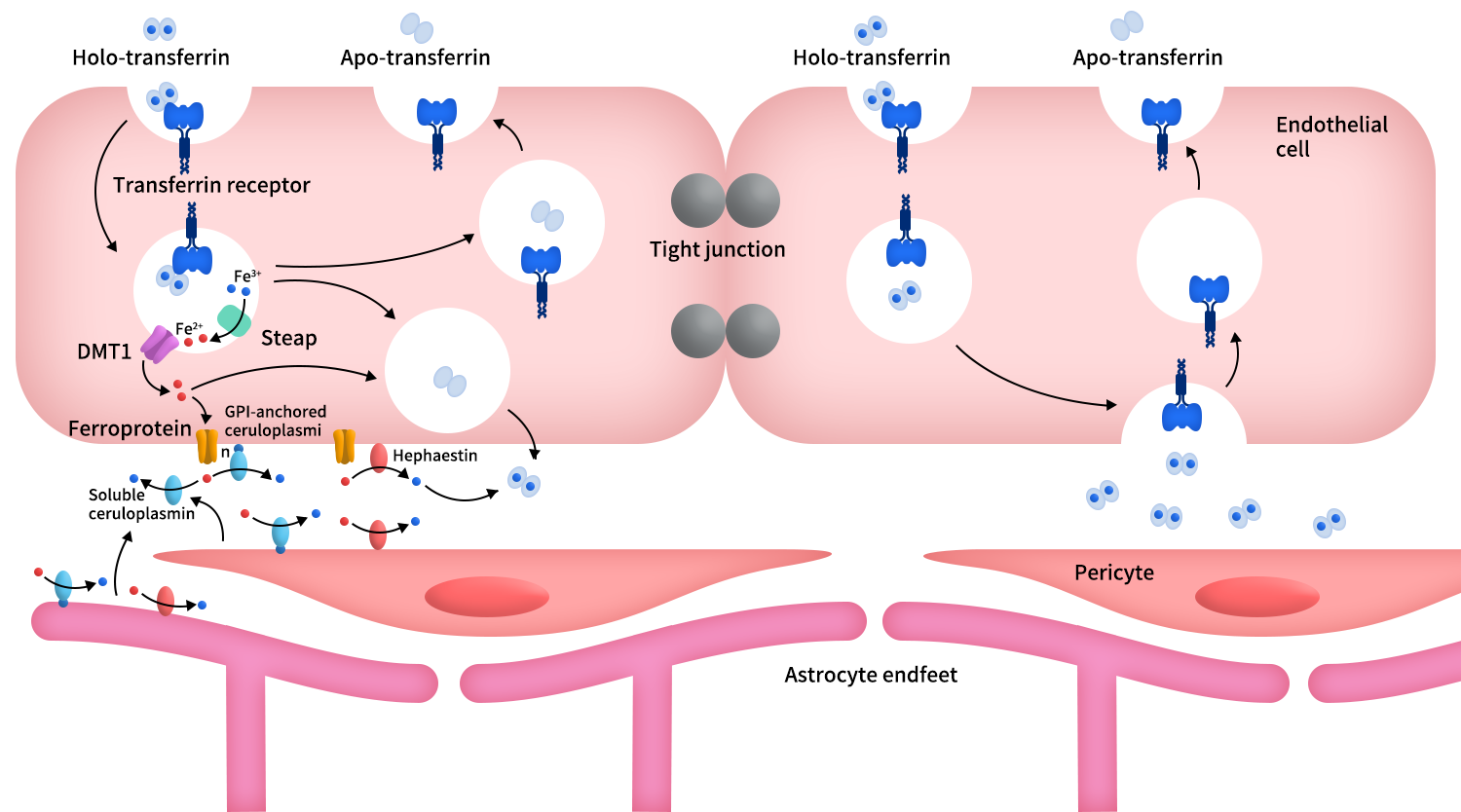
Figure 2. Schematic of transferrin receptor (TfR)-mediated transcytosis and iron handling at the blood–brain barrier (BBB) [6]
In addition to the classical iron transport pathway, TfR1 also enables receptor-mediated transcytosis. Certain ligands, such as engineered antibodies, bind to TfR1 and, instead of being degraded intracellularly, are transported across endothelial cells via vesicular trafficking and released intact on the brain side. This transcytosis mechanism avoids lysosomal degradation, making TfR1 one of the most important natural entry points for delivering biologics (e.g., antibodies, peptides) into the central nervous system [4] [5].
However, successful transcytosis is not simply a matter of binding. Studies show that transport efficiency is highly dependent on ligand design [5] [7]:
- Binding affinity: Excessively high affinity may trap the TfR1–ligand complex in lysosomes, leading to degradation; moderate affinity is generally more favorable for transcytosis.
- Valency (binding format): Monovalent vs. bivalent interactions can significantly alter receptor trafficking pathways.
- Epitope selection: Different binding epitopes on TfR1 can influence internalization routes and ultimate intracellular fate.
4. Clinical Progress of TfR1-Targeted Therapeutics
In recent years, strategies targeting Transferrin Receptor 1 (TfR1) have steadily transitioned from early-stage research into clinical validation. According to incomplete statistics, there are currently around 114 TfR1-related drug programs worldwide, including 30 in the drug discovery stage, 58 in preclinical development, 11 in clinical trials, and only 1 approved product to date.
Although the number of approved and clinical-stage assets remains relatively limited, the diversity of therapeutic modalities is notable. These include bispecific antibodies, antibody–oligonucleotide conjugates (AOC), monoclonal antibodies, and antibody–drug conjugates (ADC).
From a therapeutic application perspective, TfR1-based strategies are rapidly expanding beyond traditional boundaries. Current research and development efforts span multiple disease areas, including Alzheimer’s disease, Parkinson’s disease, brain tumors and nucleic acid drug delivery. This broad applicability highlights TfR1’s growing importance not only as a BBB transport receptor but also as a platform technology for central nervous system (CNS) drug delivery [5] [8].
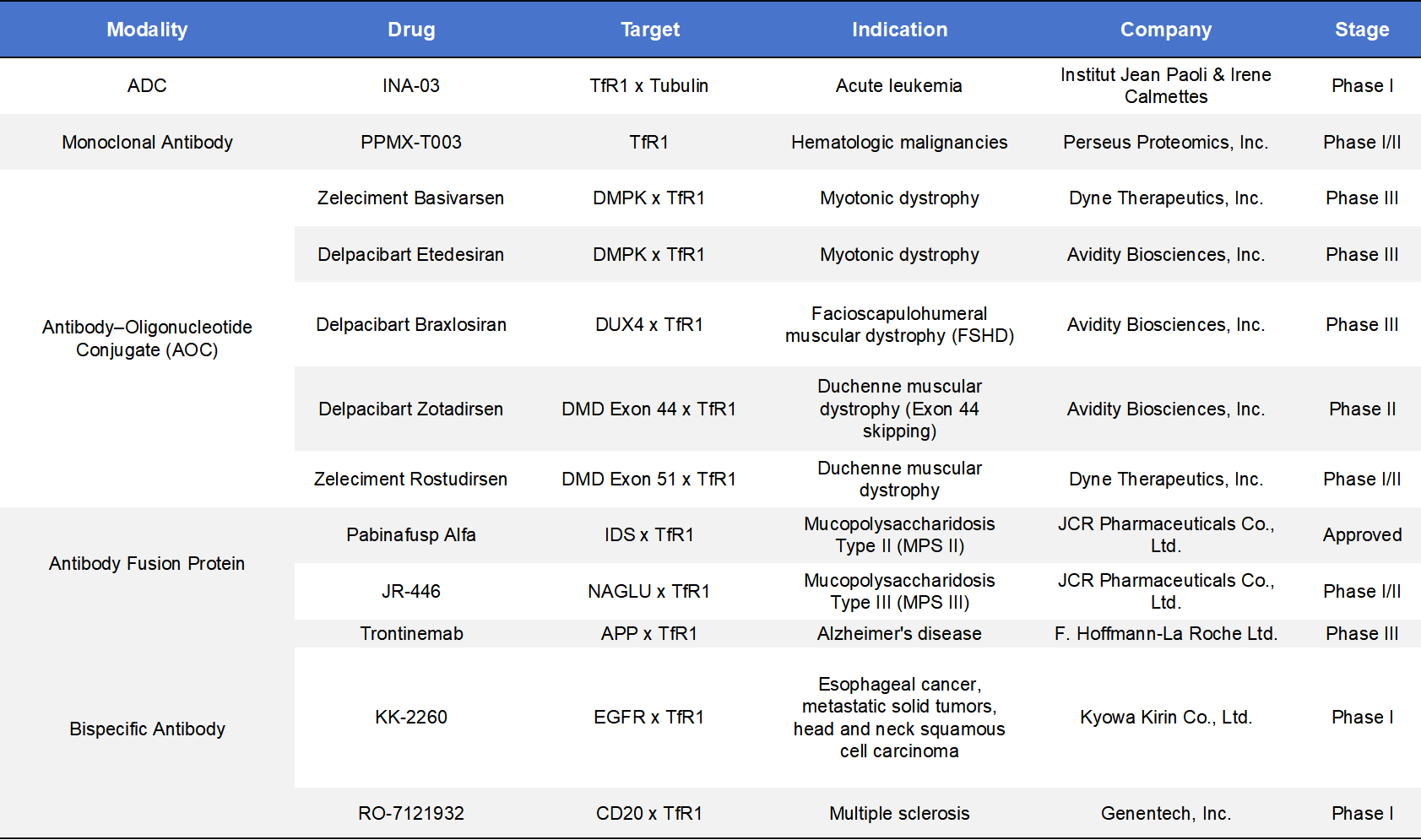
4.1 TfR1-Targeted ADCs
Currently, only one antibody–drug conjugate (ADC) targeting Transferrin Receptor 1 (TfR1) has entered clinical development: INA-03. This candidate is co-developed by Institut Paoli-Calmettes (IPC) and Inatherys and is currently in a Phase I clinical trial (NCT03957915) for relapsed/refractory acute myeloid leukemia (AML) and acute lymphoblastic leukemia (ALL).
Its mechanism of action leverages the high expression of TfR1 in rapidly proliferating tumor cells. The antibody component specifically binds TfR1 and competitively interferes with transferrin-mediated iron uptake, while promoting internalization of the receptor complex into lysosomes. Upon internalization, the cytotoxic payload MMAE (monomethyl auristatin E) is released, leading to microtubule disruption, cell cycle arrest, and tumor cell apoptosis. Early clinical data have shown reductions in bone marrow blast cells and preliminary signs of efficacy in some patients. However, due to CD71 expression in normal hematopoietic cells, treatment-related myelosuppression remains the primary dose-limiting toxicity.
4.2 TfR1 Monoclonal Antibodies
PPMX-T003, developed by Perseus Proteomics, Inc., is a human monoclonal antibody targeting TfR1. It represents a novel class of therapeutics based on modulation of the iron metabolism pathway.
Its mechanism involves binding to TfR1 and inhibiting iron uptake in tumor or abnormally proliferating cells. This disrupts proliferation of erythroid precursor cells and tumor cells by reducing iron utilization, thereby exerting anti-proliferative effects independent of traditional cytotoxic mechanisms.
Initially developed for polycythemia vera (PV), the drug has been expanded into hematologic malignancies such as aggressive natural killer-cell leukemia (ANKL). Phase I results in PV demonstrated reductions in red blood cell parameters and decreased reliance on phlebotomy in some patients, with an overall manageable safety profile. An investigator-initiated Phase I/II trial in ANKL is ongoing, although patient enrollment has been slow due to the rarity of the disease.
4.3 TfR1-Targeted AOCs
Antibody–oligonucleotide conjugates (AOCs) are a class of therapeutics that link antibodies with oligonucleotides (e.g., siRNA or antisense oligonucleotides, ASOs) via chemical conjugation. These molecules utilize antibody-mediated targeting (such as TfR1 binding) to achieve tissue-specific delivery, while the oligonucleotide component exerts gene-regulatory effects intracellularly (e.g., gene silencing or splice modulation).
In muscle diseases, TfR1 is widely used as a delivery target due to its relatively high expression in muscle cells, enabling enhanced uptake of nucleic acid therapeutics. Leading players in this space include Dyne Therapeutics, Inc. and Avidity Biosciences, Inc., both of which have developed differentiated TfR1-based delivery platforms.
- Dyne Therapeutics(Zeleciment Basivarsen & Zeleciment Rostudirsen)
Zeleciment Basivarsen: Targets DMPK for myotonic dystrophy type 1 (DM1). By conjugating a DMPK-targeting oligonucleotide with a TfR1 antibody, it enables TfR1-mediated uptake into muscle cells, reducing toxic DMPK transcripts. Currently in Phase III with promising delivery efficiency.
Zeleciment Rostudirsen: Targets DMD exon 51 mutations. Uses a TfR1 antibody–ASO strategy to enhance muscle delivery and restore dystrophin expression via exon skipping. Currently in Phase I/II.
- Avidity Biosciences(Delpacibart Etedesiran & Delpacibart Braxlosiran & Delpacibart Zotadirsen)
Delpacibart Etedesiran: Targets DMPK and delivers siRNA via TfR1-mediated uptake into muscle cells. A flagship AOC program, currently in Phase III, showing strong target knockdown and favorable safety.
Delpacibart Braxlosiran: Targets DUX4 for facioscapulohumeral muscular dystrophy (FSHD). Delivers siRNA to suppress DUX4 expression and slow disease progression. Currently in Phase III.
Delpacibart Zotadirsen: Targets DMD exon 44 mutations. Uses TfR1-mediated delivery of ASO to promote exon skipping and restore partial dystrophin expression. Currently in Phase II. Compared to traditional ASOs, this strategy significantly improves muscle uptake efficiency.
4.4 TfR1 Antibody Fusion Proteins
Antibody fusion proteins are recombinant molecules that combine functional proteins (e.g., enzymes or ligands) with antibodies or antibody fragments. Their key advantage lies in leveraging antibody targeting (such as TfR1 binding) to deliver therapeutic payloads to specific tissues, where the fused protein exerts its function.
- Pabinafusp Alfa, developed by JCR Pharmaceuticals Co., Ltd., is a fusion of iduronate-2-sulfatase (IDS) with an anti-TfR1 antibody. It is approved in Japan for mucopolysaccharidosis type II (MPS II). By utilizing TfR1-mediated transcytosis, it crosses the BBB and delivers the enzyme to the central nervous system, enabling degradation of accumulated glycosaminoglycans. It is the first commercial validation of TfR1-mediated brain delivery[8].
- JR-446, also from JCR Pharmaceuticals Co., Ltd., is a fusion of NAGLU enzyme with a TfR1 antibody for mucopolysaccharidosis type III (MPS III). It follows a similar design principle and is currently in Phase I/II, further expanding TfR1 applications in lysosomal storage disorders.
4.5 Bispecific Antibodies
Bispecific antibodies are engineered molecules capable of simultaneously binding two different targets. Typically, one arm binds a therapeutic target, while the other engages a delivery-related receptor such as TfR1, enabling both targeting and transport functions.
- Trontinemab, developed by F. Hoffmann-La Roche Ltd., is based on the Brainshuttle™ platform. It binds both amyloid-β and TfR1, enabling BBB transcytosis and increased brain exposure. It is currently in Phase III (TRONTIER 1/2) and represents one of the most advanced TfR1-based CNS delivery programs[9] [10].
- KK-2260, developed by Kyowa Kirin Co., Ltd., targets EGFR and TfR1 for solid tumors including esophageal cancer and head and neck cancers. It aims to enhance tumor distribution and potential barrier penetration while inhibiting EGFR signaling. Currently in Phase I.
- RO-7121932, developed by Genentech, Inc., targets CD20 and TfR1. It is designed to extend B-cell depletion into the central nervous system for multiple sclerosis treatment via TfR1-mediated BBB transport. Currently in Phase I.
5. TfR1-Related Products from DIMA BIOTECH
With the growing importance of TfR1 in brain delivery, high-quality research tools are essential to accelerate drug development. Dimab Bioscience provides a comprehensive portfolio of TfR1/TFRC-related products, including recombinant proteins, antibodies, stable cell lines, and biosimilar reference antibodies.
Their recombinant TfR1 proteins are expressed in HEK293 cells and focus on the extracellular domain (ECD), with options for His-tag, Fc-tag, or multivalent formats. The ECD preserves native binding sites, making it suitable for antibody screening and affinity assays (ELISA, BLI, SPR). Fc fusion or multimerization enhances stability and better mimics the native receptor state, supporting functional assays and mechanistic studies.
The product portfolio covers multiple species, including human, mouse, and cynomolgus monkey, enabling cross-species studies—particularly valuable for antibody development and BBB shuttle evaluation.
Additionally, the company offers biosimilar reference antibodies, flow cytometry antibodies, labeled antibodies, and stable cell lines, supporting FACS analysis and functional validation. Beyond TfR1, the platform extends to other BBB targets such as CD98hc, GLUT1, and IGF-1R, providing comprehensive support for CNS drug delivery research.
In addition to proteins and antibodies, DIMA BIOTECH is a biotechnology company focused on preclinical products and services for druggable targets. Leveraging its proprietary single B-cell antibody discovery platform, antibody engineering platform, and functional validation platform, the company has successfully identified multiple functional antibody candidates targeting BBB-related pathways. For more functional data and collaboration opportunities, please feel free to contact us.

Reference:
- Kawabata H. Transferrin and transferrin receptors update. Free Radical Biology and Medicine, 2019.
- Gammella E, et al. Iron metabolism in health and disease. International Journal of Molecular Sciences, 2017.
- Harms, Kristian and Thorsten Kaiser. Beyond soluble transferrin receptor: old challenges and new horizons. Best practice & research. Clinical endocrinology & metabolism 29 5 (2015): 799-810.
- Torti SV, Torti FM. Iron and cancer: more ore to be mined. Nature Reviews Cancer, 2013.
- Daneman R, Prat A. The blood–brain barrier. Cold Spring Harbor Perspectives in Biology, 2015.
- Shen, X.; Li, H.; Zhang, B.; Li, Y.; Zhu, Z. Targeting Transferrin Receptor 1 for Enhancing Drug Delivery Through the Blood–Brain Barrier for Alzheimer’s Disease. Int. J. Mol. Sci. 2025, 26, 9793.
- Pardridge WM. Blood–brain barrier and delivery of protein therapeutics. NeuroRx, 2020.
- Bien-Ly N, et al. Transferrin receptor-mediated transcytosis of antibodies. Journal of Experimental Medicine, 2014.
- Sonoda H, et al. A blood-brain-barrier-penetrating antibody–enzyme fusion protein. Nature Biotechnology, 2018.
- Roche clinical updates on trontinemab, 2025.
