In recent years, drug delivery across the blood-brain barrier (BBB) has progressed from proof-of-concept to clinical validation. In July 2025, Roche’s trontinemab demonstrated that at a dose of 3.6 mg/kg, 91% of patients achieved amyloid PET negativity after 28 weeks, with ARIA-E incidence below 5%, further validating the clinical potential of BBB shuttling strategies [1]. Meanwhile, AbbVie’s acquisition of Aliada Therapeutics explicitly incorporated BBB penetration technologies into its core deal rationale [2]. Even earlier, JCR’s pabinafusp alfa was approved in Japan, becoming a commercial benchmark in this field [3].
Together, these advances indicate that the BBB is no longer merely a development barrier but is becoming a critical gateway that defines competitiveness in CNS drug development. Before diving into BBB shuttling mechanisms, it is helpful to first understand what the BBB actually is.
1. What is the Blood–Brain Barrier (BBB)?
As illustrated, the BBB is a highly complex and dynamically regulated structure. Its core consists of brain microvascular endothelial cells (ECs) connected by tight junctions, supported by the basement membrane, astrocytes, pericytes, and extracellular matrix (ECM).
The primary functional interface lies at brain capillaries, serving as the key exchange surface between blood and brain tissue, with a total surface area of approximately 12-18 m², making it the largest blood, brain exchange interface in the body.
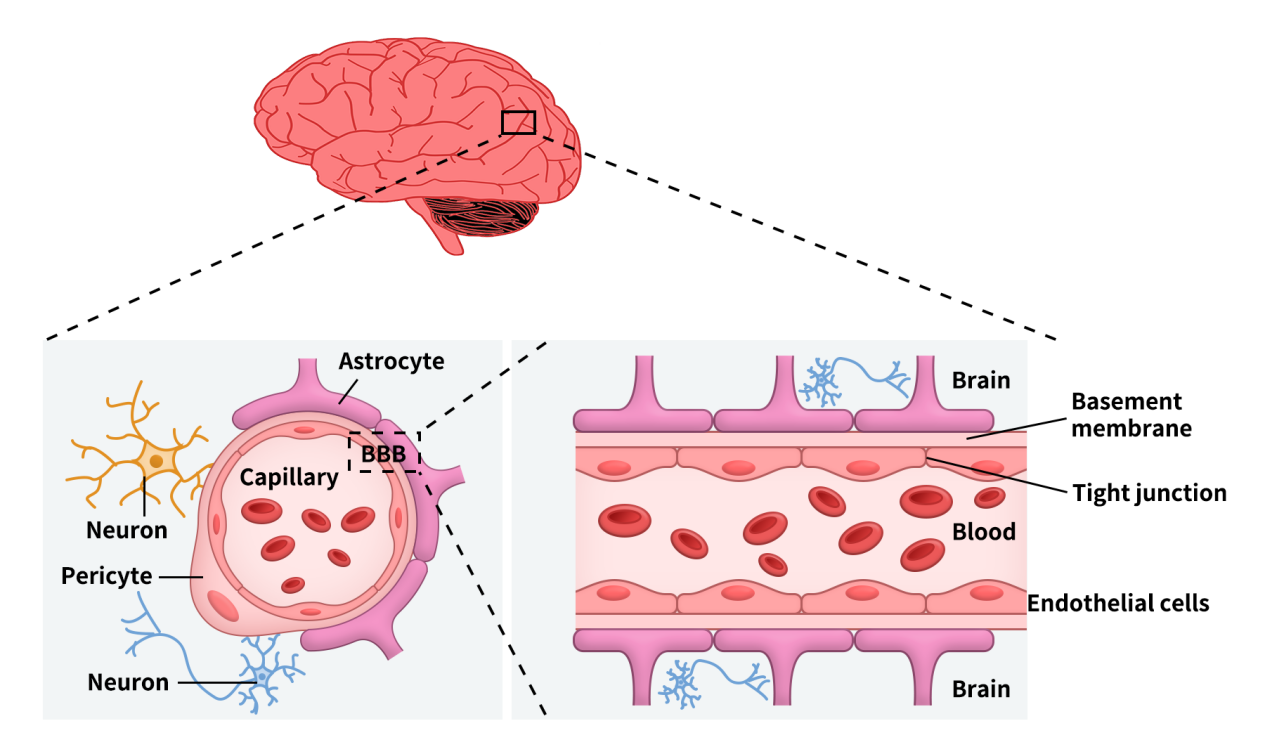
Figure 1. Blood-brain barrier (BBB) components and composition [4]
In addition to the BBB, the brain also contains the blood, cerebrospinal fluid barrier (BCSFB) and the arachnoid barrier. However, due to the lack of vasculature and relatively small surface area, their contributions to substance exchange are limited.
BBB function relies on the coordinated activity of the neurovascular unit (NVU), including endothelial cells, astrocytes, and pericytes. Astrocytes envelop capillaries via end-feet and secrete regulatory factors to maintain endothelial stability, while pericytes provide structural support and regulate permeability and blood flow. Notably, pericytes exhibit functional heterogeneity: those on the arterial side regulate blood flow, whereas those in capillary and venous regions primarily maintain barrier integrity and immune homeostasis.
It is precisely the interplay among endothelial cells, astrocytes, and pericytes that sustains the structural stability and selective permeability of the BBB.
2. Overview of BBB Shuttling Mechanisms
From a drug development perspective, the key question is no longer whether drugs can cross the BBB, but how they do so. Currently, three major pathways dominate: receptor-mediated transcytosis (RMT), carrier-mediated transport (CMT), and adsorptive-mediated transcytosis (AMT) [4,5].
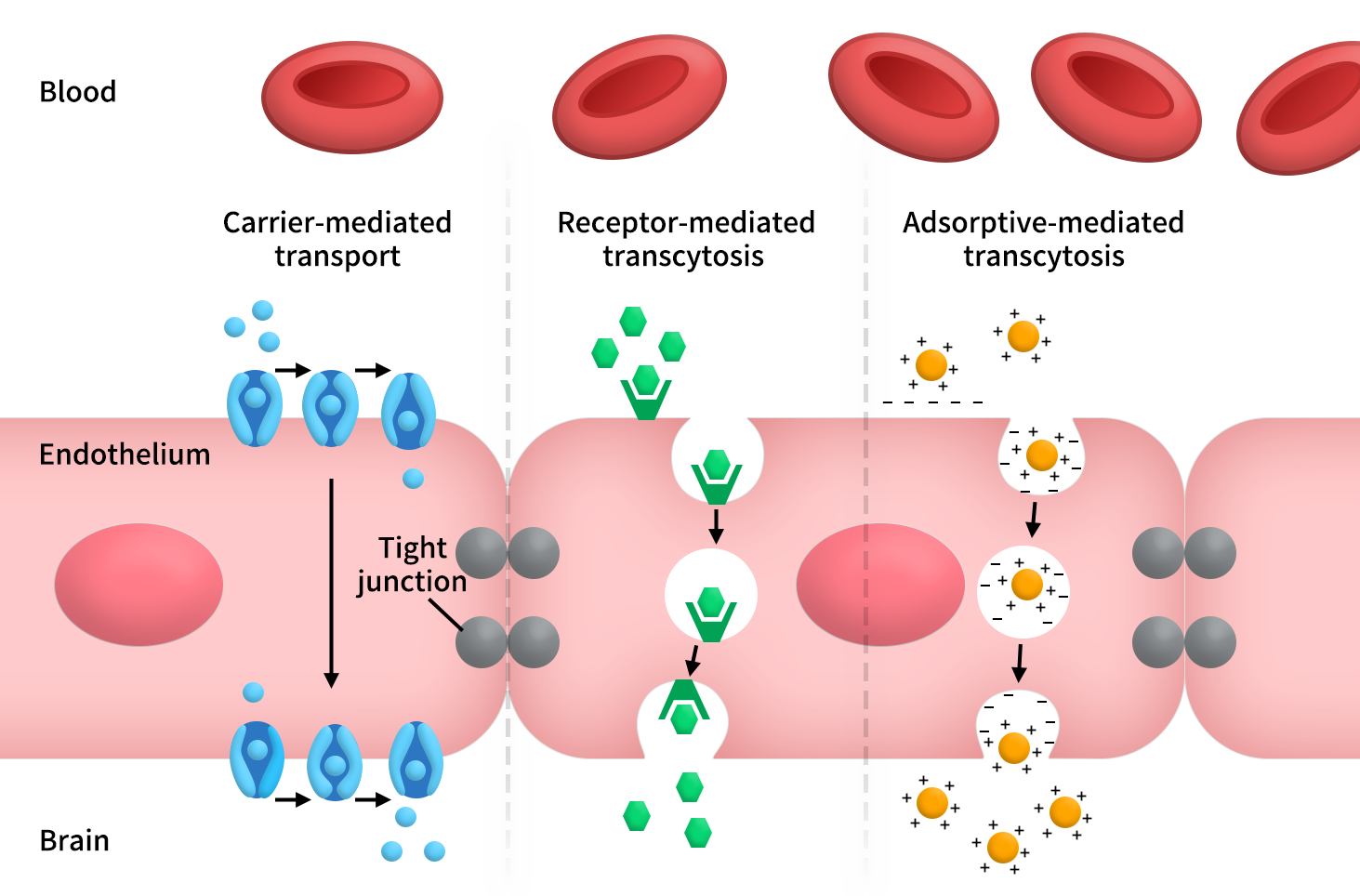
Figure 2. Three main ways through the BBB
Among these, RMT is the most mature and closest to becoming a platform technology. Its core logic is that drugs first bind to specific receptors on BBB endothelial cells, followed by receptor-mediated endocytosis, intracellular trafficking, and transcytosis into the brain parenchyma. Because this mechanism naturally accommodates large molecular cargo, modalities such as antibodies, enzyme replacement therapies, bispecific antibodies, fusion proteins, and even certain nucleic acid conjugates typically prioritize the RMT route.
In contrast, CMT is better suited for small molecules, prodrugs, and molecular mimicry strategies. It leverages endogenous transport systems for nutrients such as glucose, amino acids, and monocarboxylates, allowing drugs to “masquerade” as natural substrates to gain CNS entry.
AMT, another frequently discussed pathway, relies on electrostatic interactions between positively charged molecules (e.g., peptides, proteins, nanoparticles) and negatively charged components on the endothelial surface, thereby enhancing endocytosis and transcytosis [4,5].
Given these mechanisms, which targets are currently most promising for BBB drug delivery?
3. RMT Targets
As noted, RMT is particularly suitable for large-molecule delivery. Key targets include transferrin receptor 1 (TfR1), insulin receptor (INSR), IGF1R, LRP1, and CD98hc. The former represent classical entry points, while the latter are emerging as next-generation gateways [5].
3.1 TfR1: The Most Validated BBB Shuttling Target
TfR1, encoded by the TFRC gene, is the most widely studied and arguably most important BBB target. Its significance stems not only from high expression on brain endothelial cells but also from extensive validation spanning preclinical and clinical stages.
JCR’s pabinafusp alfa and Roche’s trontinemab both exemplify successful TfR1-mediated BBB delivery [1,3], establishing TfR1 not just as a useful receptor but as a reproducible development paradigm.
Importantly, TfR1 also highlights that BBB delivery is not as simple as binding equals crossing. Classic studies by Bien-Ly et al. demonstrated that brain uptake depends critically on antibody affinity, valency, and intracellular trafficking fate. High affinity does not necessarily improve delivery and may instead lead to receptor downregulation, intracellular retention, or lysosomal degradation [6].
3.2 CD98hc: A Rising Next-Generation Entry Point
CD98hc, encoded by SLC3A2, has rapidly gained attention. Research by Chew et al. showed that CD98hc-targeted transport vehicles achieve differentiated brain delivery kinetics in humanized mice and cynomolgus monkeys. Compared to TfR1, CD98hc exhibits slower but more sustained brain exposure, with distinct distribution and safety profiles [7].
This suggests that CD98hc is not merely an alternative to TfR1 but may represent a distinct class of delivery gateway, particularly advantageous for therapeutics requiring prolonged CNS exposure.
3.3 IGF1R/INSR: High Physiological Relevance, Higher Translational Complexity
IGF1R and INSR are also frequently explored RMT targets due to their roles in brain metabolism and growth signaling. Shin et al. reported an IGF1R-based shuttle (Grabody B) capable of safely delivering biologics into the brain using non-neutralizing antibodies [8].
However, these targets are more challenging to develop than TfR1. Their tight coupling to systemic metabolism and broad peripheral expression complicate safety, species translation, and off-target effects [5]. Their appeal lies in physiological relevance, but this is also the source of their complexity.
4. CMT Targets
CMT is primarily suited for small molecules and prodrug strategies, where success depends on molecular recognition by transporters rather than antibody engineering [4,9]. Key targets include LAT1, GLUT1, OATP, and MCT.
4.1 LAT1
LAT1 is one of the most established BBB transporters. It has been widely used for drug delivery across biological barriers, with CNS-active drugs such as L-DOPA, gabapentin, and pregabalin often cited as examples [10].
LAT1 effectively transforms BBB penetration into a medicinal chemistry problem—optimizing molecular structures to match transporter preferences. It is particularly well-suited for prodrug design, making it one of the most engineerable entry points for CNS drug development [9,10].
4.2 GLUT1
GLUT1 (SLC2A1) is the primary glucose transporter at the BBB and is essential for brain energy supply. Its importance makes it an attractive yet challenging target. While glycosylation strategies may enhance brain uptake, competition with endogenous glucose transport poses significant risks to metabolic stability and safety [11].
Thus, GLUT1 is better suited for highly constrained and precisely optimized approaches rather than general-purpose delivery platforms.
4.3 OATP and MCT
Other uptake transporters such as OATP and MCT also present opportunities. However, compared to LAT1, they are more context-dependent—often applicable to specific substrates, disease states, or drug classes rather than universal platforms [4,9].
5. AMT Pathway
AMT is more of a strategy driven by molecular properties (e.g., charge) rather than defined receptor targets. Its advantage lies in broad cargo compatibility, but its drawbacks include low specificity and higher risk of off-target distribution [4,5].
While no consensus “top-tier” targets exist, heparan sulfate proteoglycans (HSPGs), including Syndecan and Glypican families, are among the closest to being targetable entities. Positively charged molecules can bind to negatively charged glycosaminoglycans on cell surfaces, triggering endocytosis and transcytosis.
6. DIMA BIOTECH Ready-to-Use Antibody Solutions Empower BBB Drug Development
High-quality antibody resources are increasingly critical for accelerating BBB-targeted drug development. DIMA BIOTECH has established a comprehensive portfolio covering key RMT and CMT targets, including TFRC/TfR1, CD98/SLC3A2, and GLUT1/SLC2A1. These ready-to-use antibody molecules can be directly applied to BBB shuttle design and functional validation, significantly shortening early-stage development timelines.
In addition, DIMA BIOTECH provides high-quality recombinant proteins and flow cytometry antibodies targeting key drug targets. Its recombinant protein portfolio includes extracellular domain (ECD) proteins and full-length multi-pass membrane proteins, available across multiple species, tags, and biotinylation formats. For more product details, please feel free to reach out.

Reference:
- Roche. Roche presents new insights in Alzheimer’s disease research across its diagnostics and pharmaceutical portfolios at AAIC. 2025.
- AbbVie. AbbVie Completes Acquisition of Aliada Therapeutics. 2024.
- JCR Pharmaceuticals. JCR Pharmaceuticals Announces Approval of IZCARGO® for Treatment of MPS II in Japan. 2021.
- Ding L, Kshirsagar P, Agrawal P, et al. Crossing the Blood-Brain Barrier: Innovations in Receptor- and Transporter-Mediated Transcytosis Strategies. Pharmaceutics. 2025;17(6):706.
- Haqqani AS, Bélanger K, Stanimirovic DB. Receptor-mediated transcytosis for brain delivery of therapeutics: receptor classes and criteria. Frontiers in Drug Delivery. 2024;4:1360302.
- Bien-Ly N, Yu YJ, Bumbaca D, et al. Transferrin receptor trafficking determines brain uptake of TfR antibody affinity variants. Journal of Experimental Medicine. 2014;211(2):233-244.
- Chew KS, Wells RC, Moshkforoush A, et al. CD98hc is a target for brain delivery of biotherapeutics. Nature Communications. 2023;14:5053.
- Shin JW, An S, Kim D, et al. Grabody B, an IGF1 receptor-based shuttle, mediates efficient delivery of biologics across the blood-brain barrier. Cell Reports Methods. 2022;2(11):100338.
- Ronaldson PT, Davis TP. Blood-brain barrier transporters: a translational consideration for CNS delivery of neurotherapeutics. Expert Opinion on Drug Delivery. 2024;21(1):71-89.
- Puris E, Gynther M, Auriola S, Huttunen KM. L-Type amino acid transporter 1 as a target for drug delivery. Pharmaceutical Research. 2020;37:88.
- Zhu Y, Verkhratsky A, Chen H, Yi C. Understanding glucose metabolism and insulin action at the blood-brain barrier: Implications for brain health and neurodegenerative diseases. Acta Physiologica. 2025;241(2):e14283.
