In recent years, immunometabolism has become a key focus in drug discovery, highlighting the interplay between metabolic pathways and immune regulation. Among emerging targets, G protein-coupled receptor 84 (GPR84, also called EX33) has gained attention as a medium-chain fatty acid (MCFA) receptor involved in inflammatory and immune processes. Recent studies show that GPR84 enhances macrophage phagocytosis and contributes to anti-tumor immunity, while also amplifying inflammation through activation of the NLRP3 inflammasome [1,2]. In addition, GPR84 has been implicated in tumor microenvironment remodeling and responses to cancer immunotherapy [3].
However, despite clear biological functions, the clinical development of GPR84-targeted therapies remains challenging. Therefore, a systematic understanding of its structure, expression, signaling pathways, and therapeutic potential is essential.
1. Structural Characteristics of GPR84
Human GPR84 belongs to the Class A (rhodopsin-like) G protein-coupled receptor (GPCR) family. Its structure consists of seven transmembrane α-helices (TM1-TM7) connected by extracellular loops (ECL1-3) and intracellular loops (ICL1-3), forming the canonical architecture required for transmembrane signal transduction.
The ligand-binding pocket of GPR84 is located within a central cavity formed by TM3-TM7 and exhibits strong hydrophobic properties, which underlie its selectivity for medium-chain fatty acids. Structural studies reveal the presence of a hydrophobic region functioning as a “chain-length selector”, enabling precise recognition of fatty acid chain length and preventing binding of ligands that are either too short or too long. This ligand selectivity mechanism is consistent with other fatty acid-sensing GPCRs, such as FFAR1 (GPR40) and FFAR4 (GPR120), which preferentially recognize long-chain fatty acids, as well as FFAR2 (GPR43) and FFAR3 (GPR41), which respond to short-chain fatty acids. Compared with these receptors, GPR84 is uniquely tuned to medium-chain fatty acids (C9-C14), highlighting its distinct role in linking lipid metabolism to immune regulation.
A key residue, R172, stabilizes the carboxyl head group of fatty acids via electrostatic interactions, establishing a dual recognition mechanism characterized by hydrophobic tail insertion and polar head anchoring. This structural feature is essential for ligand specificity and receptor activation.
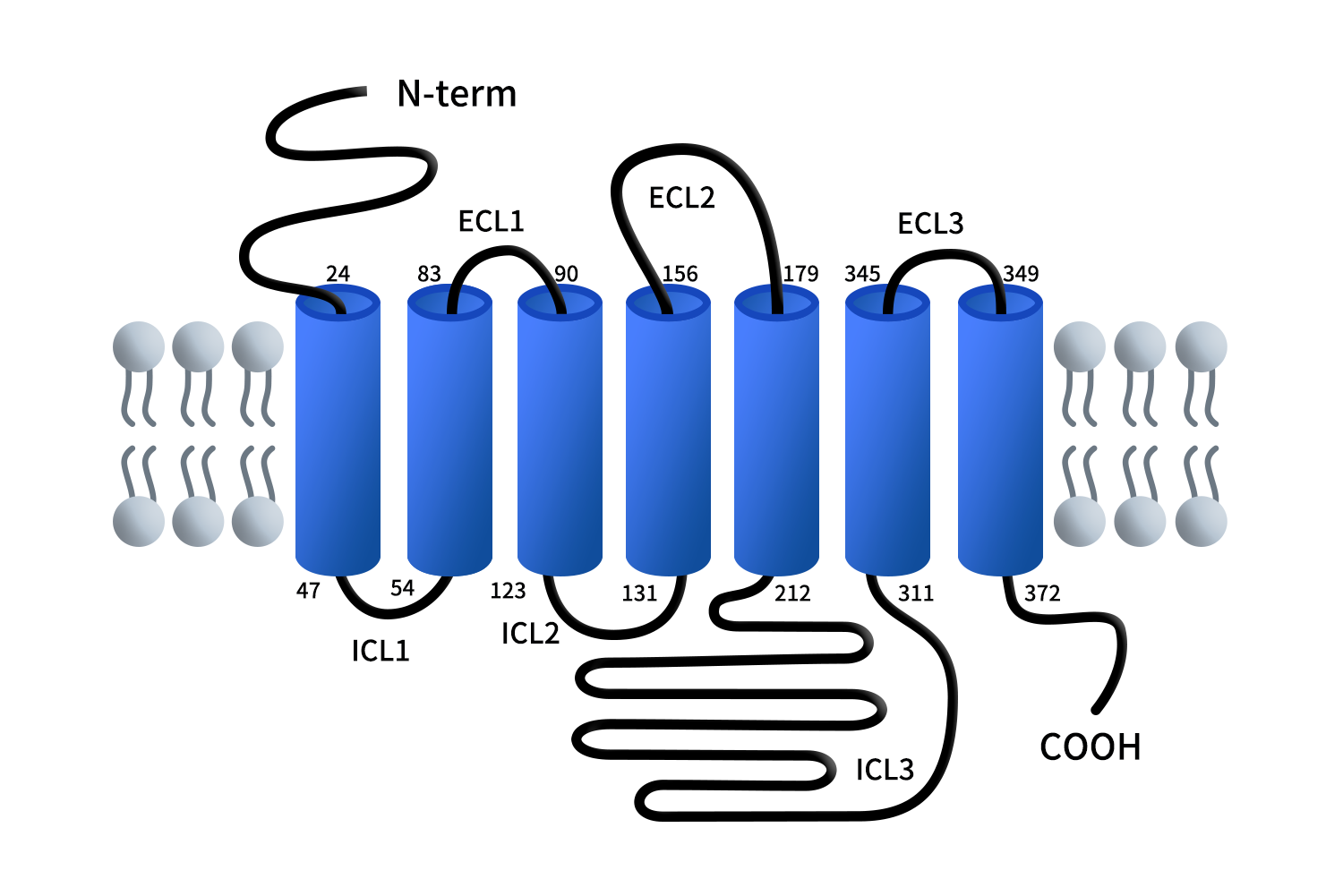
Figure 1. The structure of G protein-coupled receptor 84 (GPR84) [4]
In addition, the extracellular loop 2 (ECL2) of GPR84 exhibits a unique conformation that partially covers the ligand-binding pocket. This loop not only directly participates in ligand interaction but also forms a “gating structure”, regulating ligand access to the receptor core. Compared with many GPCRs that have relatively open ligand pathways, this structural arrangement suggests that GPR84 possesses a higher degree of ligand selectivity and binding control.
During receptor activation, GPR84 displays canonical GPCR behavior, including outward movement of transmembrane helix TM6 to create a G protein-binding interface. However, its microswitch mechanism is distinct. The highly conserved W6.48 residue found in many GPCRs is replaced by Y6.48 in GPR84, resulting in differences in hydrogen-bonding networks and conformational transitions. Consequently, receptor activation relies more heavily on hydrophobic interactions between fatty acid ligands and the binding cavity [4].
2. Expression and Physiological Functions of GPR84
GPR84 is predominantly expressed in immune cells, including neutrophils, monocytes, and macrophages, with particularly high expression levels in macrophages [5]. Beyond the immune system, GPR84 is also detected in adipose tissue, lung tissue, and the central nervous system, especially in microglia [6].
Functionally, GPR84 acts as a pro-inflammatory receptor. Its activation enhances immune cell chemotaxis, promotes the release of pro-inflammatory cytokines, and induces reactive oxygen species (ROS) production, thereby amplifying inflammatory responses [8].
In macrophages, GPR84 plays a central regulatory role. It has been shown to enhance phagocytic activity [1] and promote M1 polarization, a pro-inflammatory phenotype associated with host defense and tissue damage [2].
Importantly, as a fatty acid-sensing GPCR, GPR84 links metabolic cues to immune responses. By detecting local changes in lipid composition, it translates metabolic signals into immune activation, making it a key node in the metabolism–immunity interface [7].
3. GPR84 Signaling Pathways
GPR84 is a Gi protein-coupled receptor, with medium-chain fatty acids, such as capric acid and caprylic acid, serving as its endogenous ligands [4]. Its downstream signaling pathways are primarily centered on inflammation regulation and immune cell activation.
First, in the Gi-cAMP signaling pathway, GPR84 activation inhibits adenylate cyclase, leading to reduced intracellular cAMP levels. Since cAMP generally exerts anti-inflammatory effects, its reduction removes inhibitory constraints on pathways such as NF-κB, thereby promoting the expression and release of pro-inflammatory cytokines and enhancing immune responses.
Second, GPR84 strongly promotes activation of the NLRP3 inflammasome. Upon activation, it enhances caspase-1 activity, leading to the maturation and secretion of IL-1β and IL-18 [2]. This mechanism plays a critical role in amplifying inflammatory cascades during infection and tissue injury and is particularly relevant in diseases such as inflammatory bowel disease (IBD) and neuroinflammation.
In addition, GPR84 induces Ca²⁺ signaling, increasing intracellular calcium levels. This rapid ionic signaling regulates immune cell chemotaxis, adhesion, and phagocytosis, thereby enhancing the responsiveness of neutrophils and macrophages to inflammatory stimuli.
4. GPR84-Related Diseases
GPR84 plays a critical role across a wide range of diseases, with its biological functions closely linked to pro-inflammatory signaling, immune regulation, and metabolic pathways. Increasing evidence highlights GPR84 as a key regulator at the intersection of inflammation, fibrosis, neuroinflammation, and tumor immunity, making it an emerging target in drug discovery and therapeutic development.
4.1 Inflammatory Diseases
In IBD, GPR84 is predominantly expressed in intestinal monocytes and macrophages, where it acts as a mediator of immune activation. Upon elevated levels of medium-chain fatty acids (MCFAs) or immune stimulation, GPR84 becomes activated and enhances the NF-κB signaling pathway, leading to increased secretion of pro-inflammatory cytokines such as IL-1β and TNF-α, thereby exacerbating intestinal inflammation [2]. Preclinical studies demonstrate that GPR84 antagonists can significantly reduce inflammatory markers and alleviate colonic tissue damage, suggesting strong therapeutic potential in IBD treatment.
In addition, in acute lung injury (ALI) and infectious inflammatory conditions, GPR84 regulates neutrophil chemotaxis and infiltration, amplifying local inflammatory responses [8]. However, excessive activation may result in tissue damage, underscoring its dual role as both a driver and amplifier of inflammation.
4.2 Fibrosis and Metabolic Diseases
GPR84 also plays a pivotal role in fibrotic diseases and metabolic disorders. In models of idiopathic pulmonary fibrosis (IPF) and liver fibrosis, GPR84 expression is significantly upregulated and positively correlated with pro-inflammatory cytokine levels and collagen deposition [9].
Mechanistically, GPR84 promotes fibrosis through its pro-inflammatory activity, activating key pathways such as TGF-β/Smad signaling, which drives fibroblast proliferation and extracellular matrix production.
These findings indicate that GPR84 functions not only as an inflammation amplifier but also as a potential driver of fibrogenesis, making it a promising therapeutic target in chronic fibrotic diseases.
4.3 Neurological Diseases
Within the central nervous system (CNS), GPR84 is primarily expressed in microglia, the resident immune cells of the brain. Upon activation, GPR84 promotes the assembly of the NLRP3 inflammasome, leading to increased release of IL-1β and IL-18, and triggering pyroptosis, a form of inflammatory cell death [10].
This process contributes to the progression of neuroinflammation and brain injury, positioning GPR84 as a potential therapeutic target in neurological conditions such as stroke, traumatic brain injury (TBI), and neurodegenerative diseases.
4.4 Cancer
The role of GPR84 in cancer is context-dependent and exhibits a dual function within the tumor microenvironment. On one hand, GPR84 enhances macrophage phagocytosis, thereby improving tumor immune surveillance and contributing to anti-tumor immunity [1]. On the other hand, in certain tumor contexts, GPR84 promotes the polarization of macrophages into immunosuppressive tumor-associated macrophages (TAMs), facilitating immune evasion and tumor progression [4].
This functional dichotomy is largely determined by the polarization state of macrophages and the local metabolic environment within the tumor microenvironment. These findings suggest that therapeutic targeting of GPR84 in oncology requires precise timing, patient stratification, and combination strategies, particularly in the context of cancer immunotherapy.
Collectively, GPR84 regulates a broad spectrum of pathological processes, including inflammation, fibrosis, neuroinflammation, and tumor immunity, forming a complex and multidimensional disease network. While this highlights its strong potential as a druggable GPCR target, it also underscores the complexity of therapeutic development and the need for precision targeting strategies.
5. Advances in GPR84 Drug Development
According to incomplete statistics, there are currently approximately 30 GPR84-targeting drug candidates worldwide, including: 10 in preclinical development, 10 in the drug discovery stage and Only 4 in clinical development.
Most programs focus on small-molecule therapeutics targeting inflammatory and fibrotic diseases, reflecting the central role of GPR84 in immune-metabolic regulation. Below are four representative clinical-stage GPR84 drug candidates.
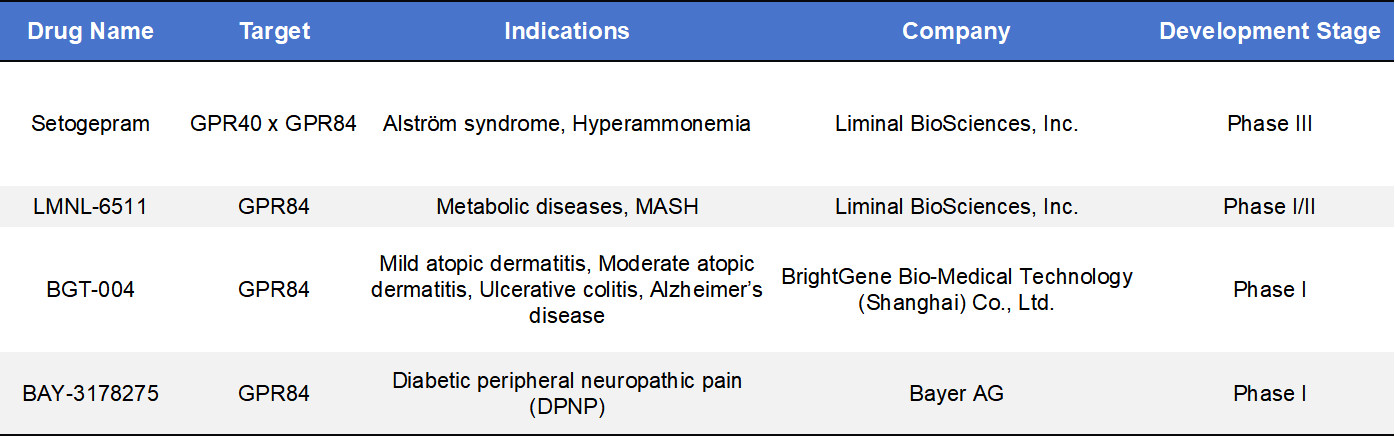
Setogepram (Fezagepras/PBI-4050) is a first-generation dual-acting modulator developed by Liminal BioSciences. It functions as both a GPR84 antagonist and a GPR40 (FFAR1) agonist, targeting multiple metabolic and inflammatory pathways. Currently in Phase III clinical trials, Setogepram has been extensively evaluated in Alström syndrome and idiopathic pulmonary fibrosis (IPF). While it demonstrated the therapeutic potential of GPR84 in anti-fibrotic treatment, its dual-target mechanism introduced pharmacological complexity. As a result, Liminal has shifted its focus toward more selective second-generation GPR84 antagonists, such as LMNL-6511.
LMNL-6511 is a novel, potent, and orally bioavailable selective GPR84 antagonist, with approximately 30-fold higher in vitro potency compared to first-generation compounds. It selectively targets GPR84 on immune cells, modulating the metabolism–inflammation axis, and is being developed for NASH/MASH, IBD, and fibrotic diseases. Currently in Phase I/II clinical trials, LMNL-6511 has demonstrated favorable pharmacokinetics (PK) in randomized, double-blind SAD/MAD studies conducted in the UK. In 2025, Liminal initiated a Phase Ib multiple-dose study in healthy subjects to further evaluate safety and tolerability. Following the acquisition of Liminal BioSciences by Structured Alpha LP in 2023, the company was taken private, with LMNL-6511 continuing as a core pipeline asset.
BGT-004 is a GPR84 antagonist with a symmetric phosphodiester structure, developed by BrightGene. Its mechanism focuses on inhibiting neutrophil infiltration and ROS production in the intestinal mucosa, thereby protecting the intestinal barrier. Preclinical studies have demonstrated strong anti-inflammatory effects in ulcerative colitis (UC) models, including improvement of colonic tissue damage. The compound is currently in Phase I clinical trials (IND approved) in China, with early-stage exploration targeting atopic dermatitis (AD) and ulcerative colitis, both immune-mediated inflammatory diseases.
BAY-3178275 is a small-molecule GPR84 antagonist developed by Bayer. Unlike other candidates primarily targeting fibrosis, this compound is being developed for neurological diseases. It modulates microglia and myeloid cell activity in the neuroinflammatory microenvironment, aiming to reduce immune-metabolism-driven neuronal damage. Currently in Phase I clinical trials (first-in-human), Bayer is evaluating its safety and preliminary efficacy in diabetic peripheral neuropathic pain (DPNP), according to its 2025–2026 pipeline disclosure.
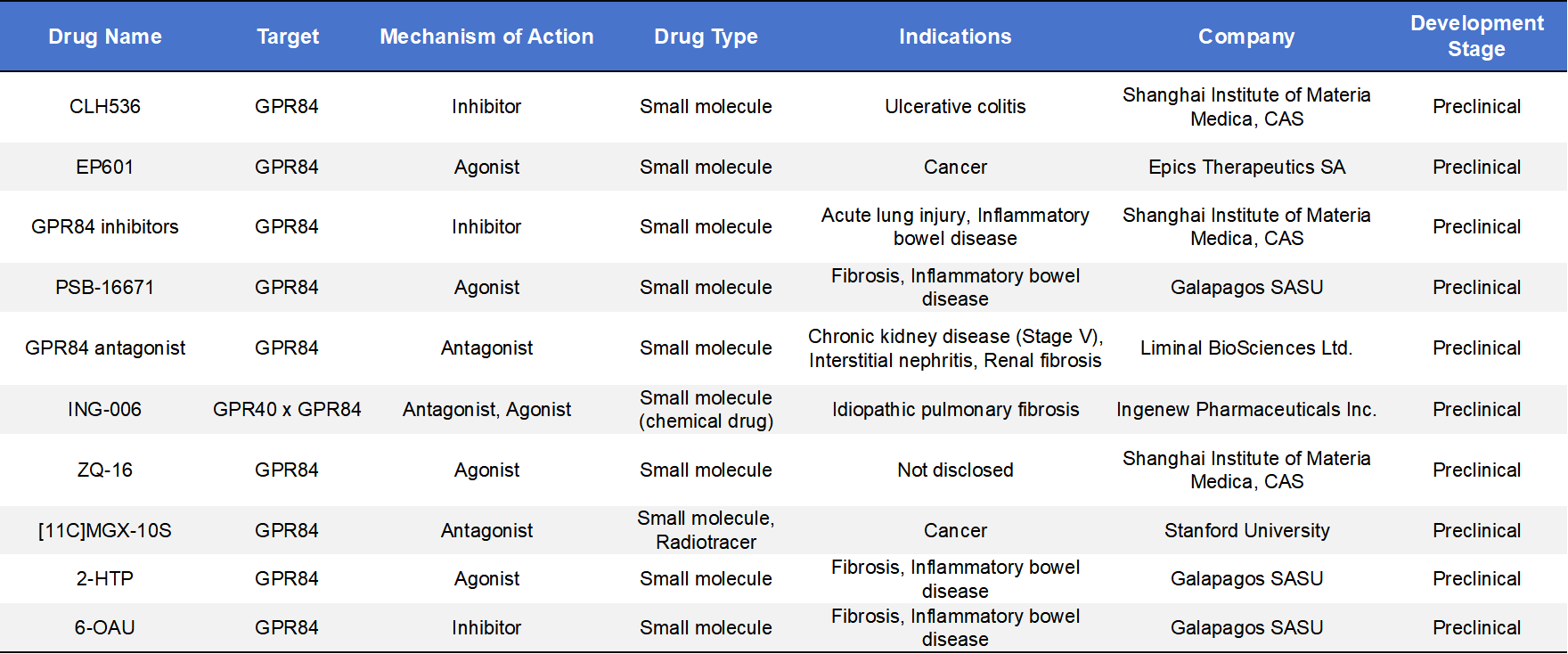
Further details of preclinical GPR84-targeted drug candidates are summarized as follows:
6.GPR84 Nanodisc Technology Enables Targeted Drug Discovery
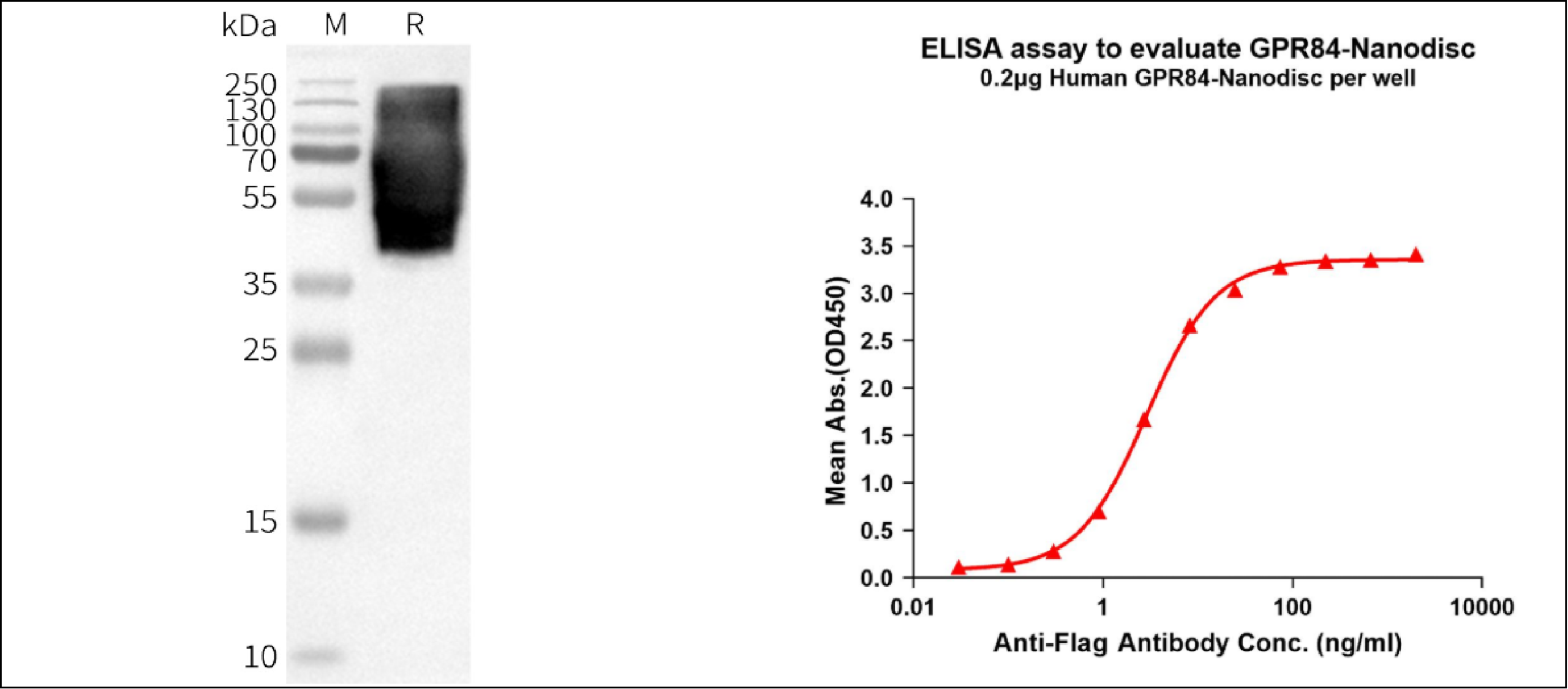
Current GPR84 drug discovery is largely focused on small-molecule development, where maintaining the native conformation of GPR84 is essential for accurate ligand screening and activity evaluation. However, due to its multi-transmembrane structure, traditional systems such as soluble fragments or crude membrane extracts often fail to preserve its structural integrity, limiting their use in binding studies and drug screening.
Full-length GPR84 nanodisc proteins, which maintain the receptor in a native lipid environment, provide a more reliable platform for mechanistic studies and lead identification. Using its mammalian expression system, Dimabio has developed full-length GPR84 nanodisc proteins with preserved native conformation and post-translational modifications in a detergent-free system. This enables more accurate structural analysis, antibody discovery, and drug–target interaction studies, making it a valuable tool for early-stage GPR84-targeted drug development.
Human GPR84 full length protein-synthetic nanodisc (FLP100129)
| Target | Cat.No. | Product Name |
| GPR84 | FLP100129 | Human GPR84 full length protein-synthetic nanodisc In Stock |
| FLP120129 | Human GPR84-Strep full length protein-synthetic nanodisc | |
| PME101470 | Human GPR84 Protein, hFc Tag In Stock | |
| FFAR1/GPR40 | FLP100134 | Human FFAR1 full length protein-synthetic nanodisc In Stock |
| FLP120134 | Human FFAR1-Strep full length protein-synthetic nanodisc | |
| FFAR2/GPR43 | FLP120236 | Human FFAR2-Strep full length protein-synthetic nanodisc In Stock |
| FFAR3/GPR41 | FLP120237 | Human FFAR3-Strep full length protein-synthetic nanodisc In Stock |
| PME101912 | Human FFAR3 Protein, hFc Tag In Stock | |
| FFAR4/GPR120 | FLP100238 | Human FFAR4 full length protein-synthetic nanodisc In Stock |
| FLP120238 | Human FFAR4-Strep full length protein-synthetic nanodisc In Stock |
References:
- Liu Y, Chen K, Wang C, et al. GPR84 regulates macrophage phagocytosis and innate immune responses. Nature Communications. 2023;14:41201.
- Wang J, Wu X, Simonavicius N, et al. GPR84 promotes NLRP3 inflammasome activation and inflammatory responses. Acta Pharmaceutica Sinica B. 2022;12(2):841-
- Zhang Q, Liu H, Chen J, et al. Targeting GPR84 reshapes tumor immune microenvironment and enhances immunotherapy. Cancer Immunology, Immunotherapy. 2023;72(11):3021-
- Schulze AS, Kleinau G, Krakowsky R, et al. Evolutionary analyses reveal immune cell receptor GPR84 as a conserved receptor for bacteria-derived molecules. iScience. 2022 Sep 6;25(10):105087.
- Audet M, Stevens RC. Emerging roles of GPR84 in inflammation and fibrosis: pharmacology and drug discovery. Frontiers in Pharmacology. 2024;15:1287654.
- Alvarez-Curto E, Milligan G. Metabolism meets immunity: the role of free fatty acid receptors such as GPR84. British Journal of Pharmacology. 2023;180(6):987-
- Nagy MA, et al. GPR84 links fatty acid sensing to immune and metabolic regulation. Journal of Clinical Investigation. 2023;133(12):e168992.
- Bouchard L, et al. GPR84 contributes to neutrophil recruitment and lung inflammation. American Journal of Respiratory Cell and Molecular Biology. 2023;69(4):456-
- Huang X, He S, Guo Y, et al. GPR84 as a potential therapeutic target in cancer and inflammation. Signal Transduction and Targeted Therapy. 2023;8:388.
- Li R, Zhao Y, Zhang M, et al. GPR84-mediated neuroinflammation and pyroptosis in CNS disorders. Journal of Neuroinflammation. 2024;21:112.
