Antibody-drug conjugates (ADCs) remain one of the most competitive and active areas in oncology drug development at AACR 2026.
Based on this year’s abstract data, the landscape is no longer a simple continuation of traditional blockbuster targets, but instead shows three clear trends. First, HER2, TROP2, and EGFR continue to dominate as the most high-frequency ADC targets, supported by both mature clinical assets and next-generation innovations such as bispecific ADCs and dual-payload ADCs. Second, ADC targets are rapidly expanding toward differentiated biology and hard-to-treat solid tumors, including NECTIN4, B7-H3 (CD276), FOLR1, c-Met, CDH17, DLL3, and CLDN18.2. These reflect both deeper validation of established targets and increasing entry of new players. Third, platform innovation is becoming more important than target selection itself, with increasing focus on payload design, linker chemistry, dual-target strategies, bystander effects, resistance reversal, and immune synergy.
From abstract frequency, the top ADC targets at AACR 2026 rank approximately as follows: HER2 (64) > TROP2 (33) > EGFR (29) > PD-L1 (18) > NECTIN4 (17) > B7-H3/CD276 (15) > FOLR1 (12) > c-Met (11) > CDH17 (10) > DLL3 (8) > CLDN18.2 (7). Below is a concise overview of these key ADC targets.
1. HER2 / ERBB2: The Hottest ADC Target at AACR 2026
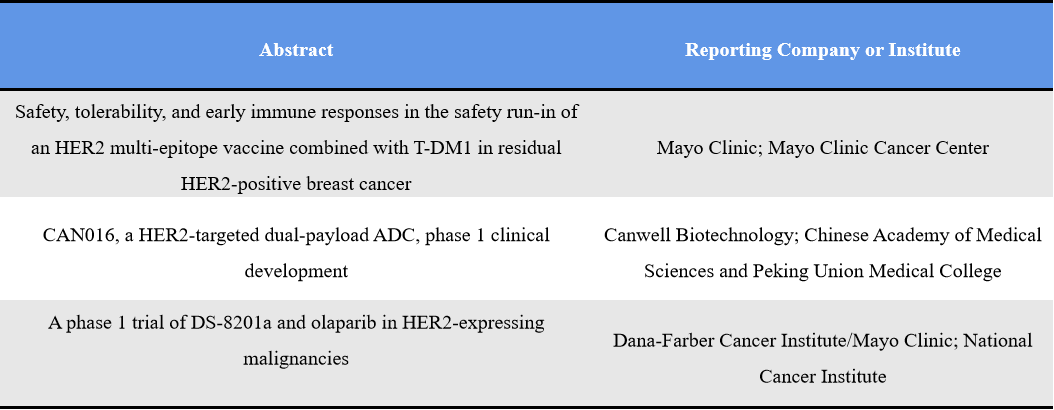
Human epidermal growth factor receptor 2 (HER2) remains the most dominant ADC target, with significantly higher activity than other targets. As one of the most clinically validated and commercially successful ADC targets, HER2 continues to evolve beyond monotherapy into HER2-low populations, combination strategies (e.g., PARP inhibitors, immunotherapy), bispecific ADCs, and dual-payload ADCs. This indicates that HER2 has transitioned from a “validated target” to a “design-driven innovation space,” where differentiation depends on payload, combination approaches, and expansion into HER2-low settings. Representative abstracts include:
2. TROP2: Expanding Beyond Breast Cancer into Broad Solid Tumors
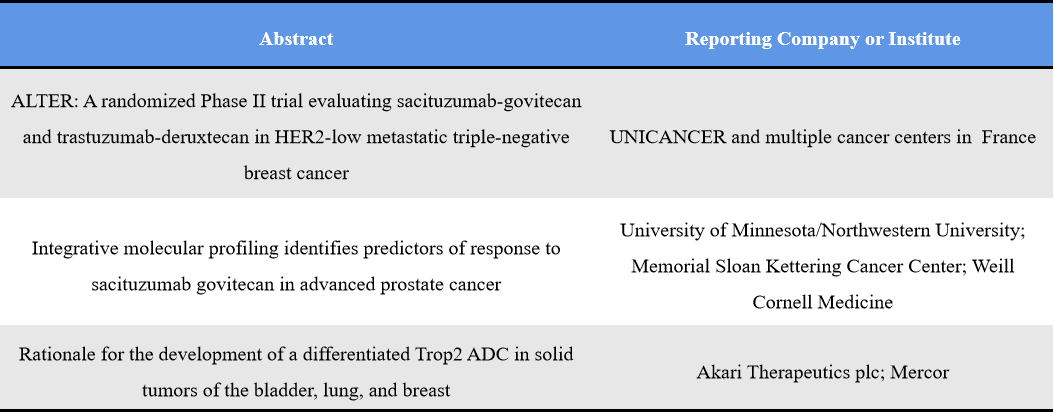
TROP2 is the second most active ADC target at AACR 2026. Following the clinical success of sacituzumab govitecan, the field is now evolving in two directions: expansion of approved molecules into additional indications and combination regimens, and development of next-generation differentiated TROP2 ADCs addressing depth of response, bystander effect, and resistance. The competition has shifted from “having a product” to “building a better product,” with emphasis on differentiation and post-resistance strategies. Representative abstracts include:
3. EGFR: A Classic Target Revitalized by ADC Technology
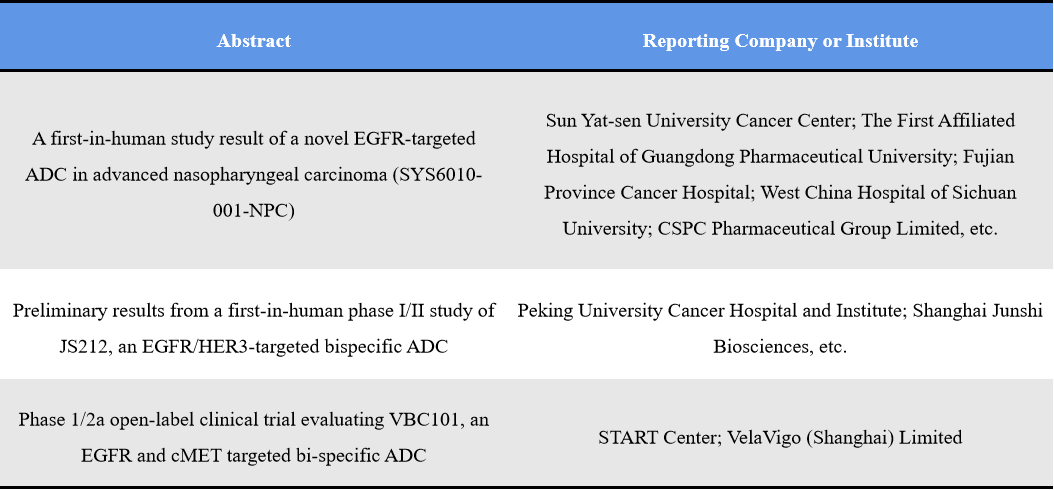
EGFR ranks as the third most prominent ADC target. Unlike traditional EGFR antibodies or TKIs, ADC technology provides new design flexibility, particularly in nasopharyngeal cancer, head and neck cancer, lung cancer, and pan-solid tumor settings. The field is rapidly evolving toward bispecific ADCs such as EGFR/HER3 and EGFR/c-Met, reflecting efforts to overcome tumor heterogeneity and resistance mechanisms. Representative abstracts include:
4. PD-L1: Immune Checkpoint Targets Enter the ADC Space
Although not traditionally a major ADC target, PD-L1 shows strong presence at AACR 2026. This reflects the convergence of ADCs and immuno-oncology, where PD-L1 is being explored not only as an immune checkpoint but also as a payload delivery target, especially in immune-cold tumors and combination therapies. Representative abstracts include:

5. NECTIN4: From Urothelial Cancer to Broader Indications
NECTIN4 remains a high-interest ADC target, with enfortumab vedotin establishing a clinical benchmark. Current development trends include next-generation NECTIN4 ADCs, strategies addressing resistance to EV, bispecific NECTIN4/TROP2 designs, and expansion into new indications such as ovarian cancer. NECTIN4 is transitioning from a single success story to a scalable platform opportunity. Representative abstracts include:

6. B7-H3 (CD276): A Pan-Solid Tumor ADC Target Gaining Momentum
B7-H3 continues to be one of the most promising next-generation ADC targets. Its broad expression across solid tumors makes it highly suitable for ADC development. The field has moved beyond initial validation into bispecific strategies, indication stratification, and mechanism optimization, reflecting increasing competition and maturity. Representative abstracts include:

7. FOLR1 (FRα): Expanding Beyond Ovarian Cancer
FOLR1 remains a highly relevant ADC target, with ongoing clinical progress and diversified molecule design. Importantly, its application is expanding beyond ovarian cancer into lung adenocarcinoma and other solid tumors, supported by increasingly diverse ADC platforms. Representative abstracts include:
8. c-Met: A Key Partner in Combination and Bispecific ADCs
c-Met represents a typical “combination-driven” ADC target. While it has standalone therapeutic value, its significance is amplified in dual-target ADCs, particularly with EGFR, addressing resistance pathways and bypass signaling. This reflects a broader strategy focused on overcoming tumor heterogeneity. Representative abstracts include:

9. CDH17: An Emerging ADC Target in Gastrointestinal Cancers
CDH17 is one of the most notable emerging targets at AACR 2026, primarily focused on gastrointestinal malignancies. Although its frequency is lower than HER2 or TROP2, its biological relevance and early clinical activity suggest strong potential as a next-generation ADC target in GI cancers. Representative abstracts include:

10. DLL3 and CLDN18.2: Two Distinct but Important ADC Directions
DLL3 continues to gain traction in neuroendocrine tumors and small cell lung cancer, representing deepening ADC development in these areas. Representative abstracts include:

Meanwhile, CLDN18.2 is expanding from monoclonal antibodies and bispecifics into ADC formats, particularly in gastrointestinal cancers. Both targets highlight diversification of ADC strategies across tumor types and mechanisms.Representative abstracts include:

In summary, the ADC landscape at AACR 2026 can be defined as follows:
- established targets are becoming increasingly competitive, emerging targets are rapidly filling the pipeline, and true differentiation is shifting toward platform innovation.
- HER2, TROP2, and EGFR remain first-tier targets, where success depends on superior design rather than target novelty. Meanwhile, B7-H3, NECTIN4, FOLR1, c-Met, CDH17, DLL3, and CLDN18.2 form the second growth wave, covering key indications such as urothelial cancer, gynecologic cancers, lung cancer, gastrointestinal tumors, and neuroendocrine malignancies.
- Notably, bispecific ADCs, dual-payload ADCs, and immune-targeting ADCs are increasing significantly, signaling that industry competition has shifted from “target discovery” to “how to design better ADCs.”
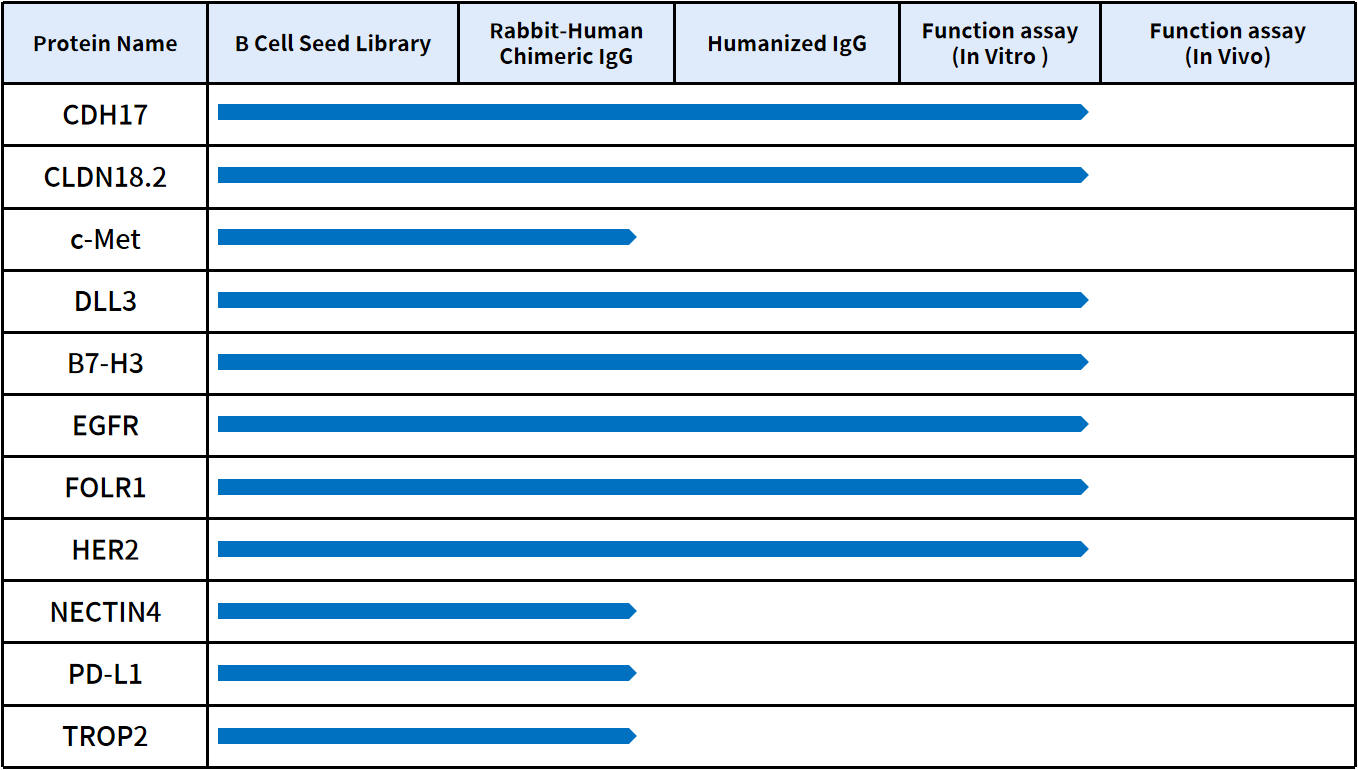
11. Accelerate ADC Drug Development with DIMA BIOTECH’s Ready-to-Use Antibody Sequences
DIMA BIOTECH is a biotechnology company focused on preclinical research solutions for druggable targets. Leveraging its proprietary single B cell antibody discovery platform, DimaBio has generated 5,000+ antibody sequences covering 500+ therapeutic targets. For selected high-interest targets, cross-reactivity with human and cynomolgus monkey proteins has been validated, along with ADC internalization and cytotoxicity data. Researchers can access antibody molecules as early as the next day for functional evaluation, significantly accelerating ADC drug development timelines. For detailed datasets and collaboration opportunities, please contact us.